Patent ductus arteriosus (PDA) : medical treatment and indications for surgical closure
Objectives
This guideline has been developed for medical staff caring for premature infants in the West of Scotland. The aim is to provide standardised PDA assessment, the rationale behind different treatment approaches and drug dosing regimes for those considering pharmacological closure of the PDA.
Closure of the DA is a normal physiological process which occurs within the 1st 24 hours in most term babies. Increasing arterial PaO2 in combination with decreasing prostaglandin levels from the placenta and secretion of bradykinin by the lung interstitium leads to circular smooth muscle contraction in the wall of the DA. This leads to relative ischaemia of the vessel wall and necrosis. Ultimately, the DA evolves into the ligamentum arteriosus (1).
This process is less efficient in preterm neonates leading to a failure of closure of the DA (‘Patent Ductus Arteriosus’ - PDA). As pulmonary pressures drop after delivery and systemic pressures increase, PDA leads to a persistent left to right shunting of blood from the high pressure aorta to the lower pressure pulmonary artery. The problems of this are two-fold:
- Increased pulmonary blood flow:
Chronic pulmonary overcirculation leads to increased pulmonary venous pressure. This leads to leakage of lymphatic fluid into the interstitium causing pulmonary oedema, changes in pulmonary mechanics and higher ventilatory requirements (2). In severe cases, pulmonary haemorrhage can develop (3).
- Decreased systemic blood flow:
‘Ductal steal’ is the concept of the PDA redirecting a proportion of systemic blood flow and diverting it to the lungs when pulmonary vascular resistance falls below systemic vascular resistance. Reduced and reversed diastolic blood flows are demonstrated by Doppler US studies in the cerebral and gastrointestinal vascular beds and there are associations between PDA and NEC, IVH, and hypotension and death (4). Closing the PDA reduces pulmonary oedema and cardiac failure but it is not clear if intervention alters other major morbidities.
The incidence of PDA increases with decreasing GA at birth, RDS, ventilation requirements, sepsis and lack of antenatal steroids (5). The rates of early closure < 28 weeks gestation, by 1 week of post natal age, are reported as very low (30% of < 28 weeks and 20% of < 26 weeks) with the likelihood that the PDA will then remain open for an average of 8 weeks (9)
Closure of the DA remains controversial. Older studies demonstrate short term benefits from closure (16) and also established the preference for medical over surgical closure, because of a better safety profile (27). There have been many studies looking at the timing of treatment as well as different regimes. Prophylactic pharmacological treatment has been shown to reduce the need for surgical closure and decrease significant pulmonary haemorrhage and IVH (7,8,25,26) but has not been shown to improve medium or long term outcomes. However there have been no RCTs exploring the impact of medical closure versus much later spontaneous closure in preterm infants. Due to lack of evidence of benefit, there are concerns that adverse effects from the pharmacological treatments may outweigh the benefits of treating the condition(6). There is, however, concern that increasing clinical tolerance of PDA in very preterm infants may result in increasing short term morbidities, fluid and nutritional restriction, increased rates of late surgical PDA ligation, increased rates of NEC and potentially adverse long term developmental outcomes.
Current management strategies are generally focussed on infants < 29 weeks gestation and include the options of pharmacological closure, conservative or supportive management (diuretics, non invasive respiratory support), surgical ligation and late catheter closure. This guideline deals with pharmacological closure. Surgical management is reserved for cases where medical closure has failed or is contraindicated and where there is inability to wean from ventilation, persisting inotrope requirement, renal failure, evidence of left ventricular damage or persistent cardiac failure near term. (See WoS MCN Guideline – Surgical Ligation of the PDA and Appendix 2). Late catheter closure is currently possible at RHC, Glasgow from a minimum weight of 6 kg. Catheter closure at lower weights have been avoided because of morbidities related to the vascular access and device deployment.
SUMMARY 1- The prevalence of PDA is high in the preterm population <29 weeks (70% at the end of week 1)- PDA causes a large left to right shunt with impaired systemic perfusion as well as pulmonary over circulation. - The presence of PDA in the preterm infant has been associated with significant morbidities, including increased rates of chronic lung disease, hypotension, pulmonary haemorrhage, IVH, NEC and mortality but there is currently little RCT evidence to support treatment- There has never been a RCT to examine whether babies do better having their PDAs closed or simply left alone.
Clinical signs. Despite evidence of a large PDA on echo from day 1, (11) clinical signs may not appear until day 4 of life and become much more pronounced as pulmonary vascular resistance falls. The following clinical signs should increase suspicion of a significant PDA:
- Bounding pulses
- Active praecordium
- Continuous murmur
- Wide pulse pressure.
Other features/ symptoms that should raise suspicion of a clinically significant PDA include:
- Inability to wean from the ventilator
- Persistently high O2 requirements
- Persistent hypotension
- Persistent or recurrent renal impairment
- Significant pulmonary haemorrhage
- Feed intolerance/ NEC
- Reversed end-diastolic flow on ultrasound Doppler in either in the cerebral circulation or superior mesenteric artery.
Chest X-ray is an important part of the assessment of any baby with PDA but has limitations as changes are affected by ventilation strategy and can be difficult to distinguish from RDS or pneumonia. Changes on x-ray associated with left heart volume overload include:
- Enlarged heart
- Bilateral lung field haziness associated with pulmonary oedema
- Air bronchograms
We do not advocate treating a PDA without confirmation of a persistent DA and an otherwise structurally normal heart by cardiac echo.
Echocardiographic findings
Assessing a PDA using echocardiography is not straightforward as there is no single measurement which accurately reflects the haemodynamic significance of a PDA. A full assessment is required, including the impact on systemic and pulmonary blood flow, which must be assessed in conjunction with the baby’s clinical condition. In addition to establishing structural normality we would encourage at least 3 measures from the selection in Table 1 (22) and an attempt should be made to grade any PDA as small, moderate or large. It is worth noting that many moderate PDAs will have pulmonary to systemic blood flow of > 2:1.
Table 1: Echocardiographic assessment of PDA
|
Feature quantified |
Small |
Moderate |
Large |
|
Characteristic of the ductus arteriosus |
|
|
|
|
Ductal Diameter (mm) |
<1.5 |
1.5-3.0 |
>3.0 |
|
Ductal Velocity Vmax (m/s) |
>2.0 |
1.5-2.0 |
<1.5 |
|
Pulmonary overcirculation |
|
|
|
|
LA:Ao Ratio |
<1.4:1 |
1.4-1.7:1 |
>1.7:1 |
|
Left PA: diastolic flow velocity (m/s) |
<0.3 |
0.3-0.5 |
>0.5 |
|
Transmitral Doppler E/A wave ratio |
<1 |
1-1.5 |
>1.5 |
|
Systemic hypoperfusion |
|
|
|
|
Descending Ao: % diastolic flow retrograde |
<30 |
30-50 |
>50 |
|
Anterior cerebral or superior mesenteric artery Diastolic blood flow pattern |
Normal (antegrade) |
Absent |
Reversed |
Cardiology assessment is recommended for persistently large PDAs, particularly in the presence of mitral valve regurgitation.
Figure 1: Doppler showing reversed end diastolic flow in the anterior cerebral artery
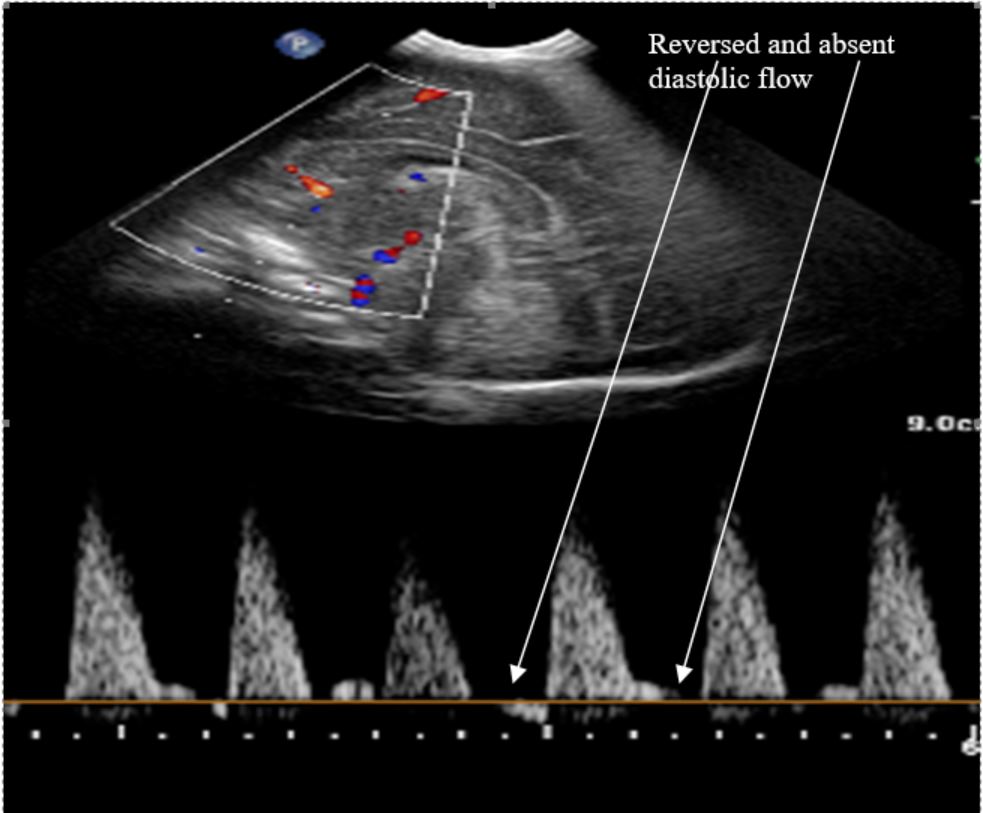
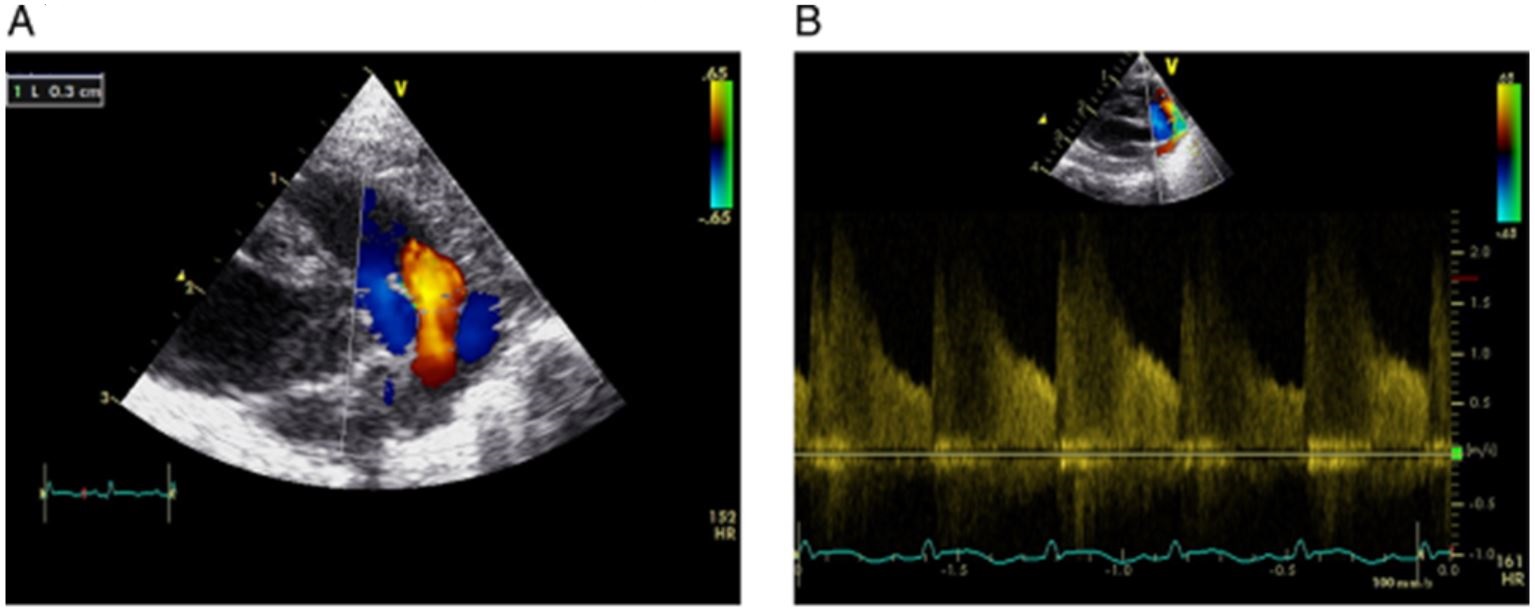
Figure 2: Echo evidence of PDA
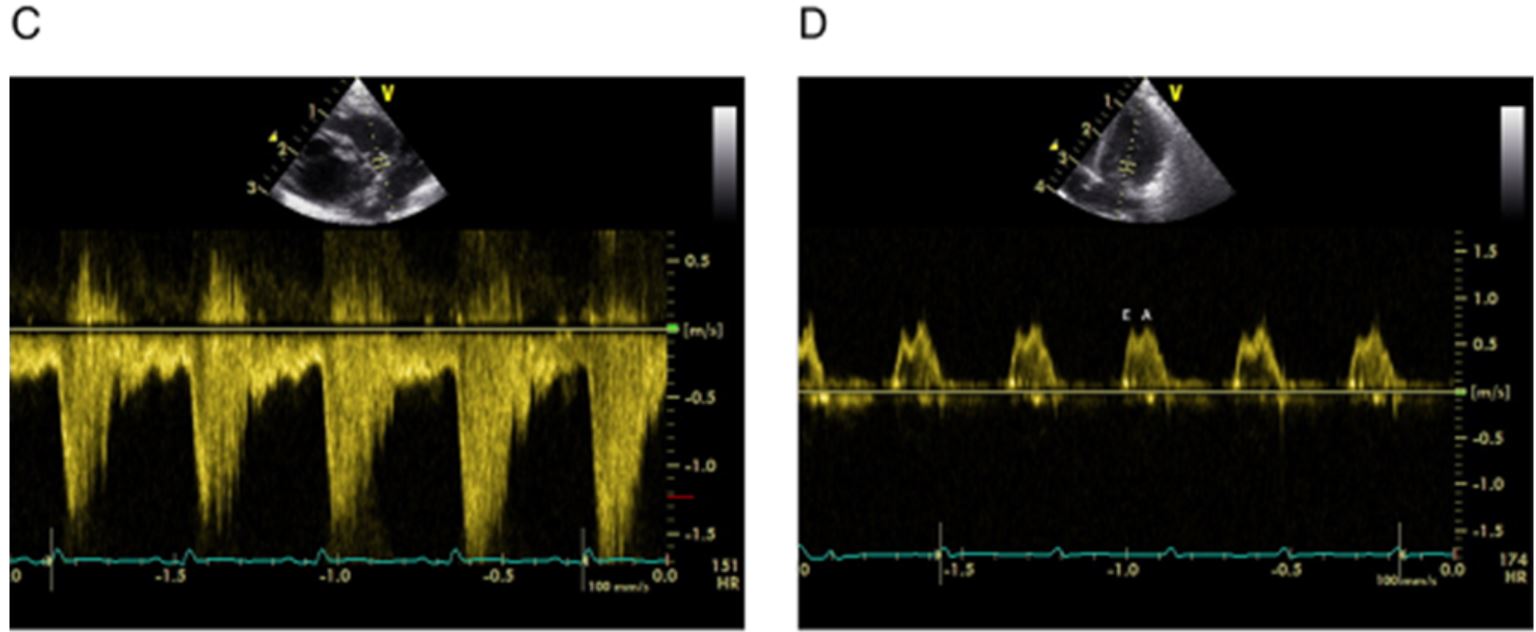
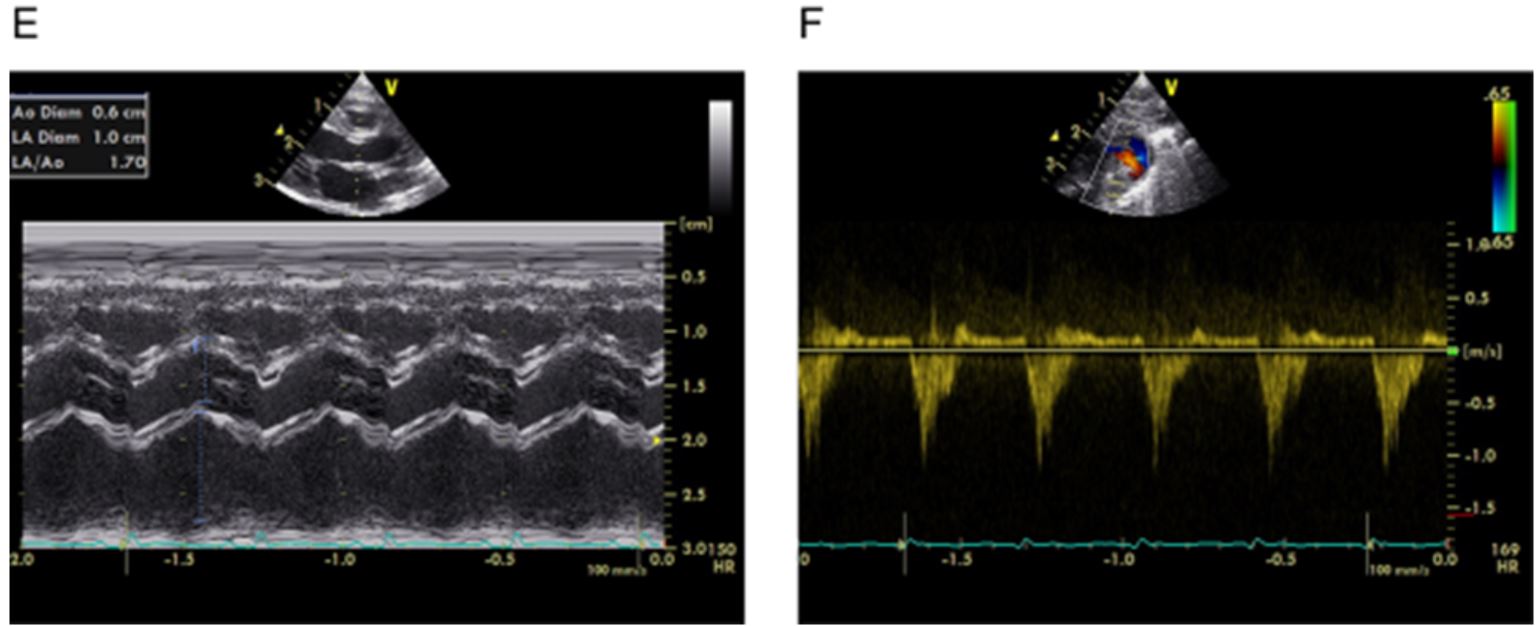
Echocardiographic assessment of haemodynamic significance of the ductus arteriosus (HSDA).29 (A) Transductal diameter: 3.0 mm=large patent ductus arteriosus (PDA) (small <1.5, moderate 1.5–3.0, large >3.0 mm). (B) Ductal velocity Vmax: 1.5–2.0 m/s=moderate PDA (small >2.0, moderate 1.5–2.0, large <1.5 m/s). (C) Antegrade left pulmonary artery (PA) diastolic flow: 0.3–0.5 m/s=moderate PDA (small <0.3, moderate 0.3–0.5, large >0.5 m/s). (D) Transmitral Doppler E/A wave ratio: approaching 1.0=moderate PDA (small <1, moderate 1–1.5, large >1.5). (E) Left atrial:aortic (LA:Ao) ratio: 1.7=large PDA (small <1.4:1, moderate 1.4±1.6:1, large >1.6:1). (F) Retrograde diastolic flow (%) in descending aorta: >50% =large PDA (grading: small <30%, moderate 30–50%, large >50%).(Heuchan and Clyman reference 9)
Cyclo oxygenase (COX) inhibitors (e.g. ibuprofen and indomethacin) or peroxidase (POX) inhibitors (paracetamol) reduce PGE 2 synthesis and result in DA smooth muscle contraction. The DA gradually becomes less PGE2 sensitive and therefore the efficacies of these medications reduce with time (9). Closure rates as low as 30% have been reported if used after the 1st week of life in the most immature populations.
Ibuprofen and indomethacin have been extensively studied. Both potentially have COX mediated side effects of thrombocytopenia, renal impairment, intestinal perforation, transiently reduced mesenteric blood flow and, in indomethacin’s case, reduction in cerebral blood flow if given as a rapid bolus. Generally both are well tolerated if given early in life.
Ibuprofen has less effect on serum creatinine levels than indomethacin although in both cases these are likely to be transient and reversible with no significant clinical impact. Ibuprofen has been reported to be associated with a significantly lower incidence of NEC compared with indomethacin in a Cochrane review(11) but notably the population receiving ibuprofen in the review studies were considerably more mature than those receiving indomethacin (11). There are a few reports of pulmonary hypertension with very early “prophylactic” ibuprofen administration. These reports are not consistent across all trials but, as there are no clear benefits to ibuprofen prophylaxis or indication for very early clinical use, this is of little significance. Concerns remain about ibuprofens efficacy at lower gestations; whilst higher doses may improve efficacy these are not routinely recommended at present because of concerns about displacement of bilirubin from albumin. However, as ibuprofen clearance increases rapidly after the 1st week of life, longer courses of ibuprofen may be beneficial when there isn’t complete echocardiographic closure after 3 doses. Ibuprofen can be administered orally, and possibly has increased efficacy by the enteral route because of a longer half life, but further study is required.
Indomethacin probably has the greatest efficacy. There are several dosing regimes described for indomethacin based on the timing of treatment again related to reductions in half life with increasing postnatal age. Shorter courses may have less association with adverse outcomes, particularly NEC and therefore the need for a longer course should always be reviewed echocardiographcally (28). Recently, there has been difficulty in obtaining indomethacin due to manufacturing issues but it is becoming increasingly available.
Paracetamol has been the subject of many recent case reviews and case series but has not been as extensively studied as ibuprofen or indomethacin. It may be a useful alternative or adjunctive therapy given its relative short term safety profile (9). There is however a lack of long term outcome data for babies treated with paracetamol for PDA.
Full dosing details, contraindications and blood monitoring are provided in the Appendices with links to the individual drug monographs.
General considerations
When considering pharmacological PDA closure, it is important that treatment should not be given at the same time as systemic corticosteroids because of the increased risk of spontaneous intestinal perforation (SIP). When considering pharmacological closure, a logical approach would be to assess the PDA from the middle to end of the first week of life in order to maximise the chances of spontaneous closure. This would also likely avoid concurrent early steroid treatment for hypotension and also avoid the commonest window for SIP. If steroids for ventilator dependence are considered likely it may be advisable to treat the PDA before commencing further steroids. Additionally, if there is evidence of cardiac failure, consideration should be given to the use of diuretics and avoidance of excessive fluid volumes.
Prophylactic treatment:
Prophylactic treatment of the PDA (on day 1) leads to a significant reduction in symptomatic PDA and later PDA ligation. In addition, rates of severe IVH, periventricular leukomalacia and pulmonary haemorrhage in the least mature infants have been reduced in studies using indomethacin prophylaxis. These findings have not been demonstrated to improve long term outcome although the studies were not powered sufficiently to assess this outcome (7). Prophylactic treatment of the PDA leads to some unnecessary exposure to potentially harmful drugs so is not routinely recommended. Although we do not routinely encourage this approach, it may be considered in some situations; particularly when there is a high risk of IVH.
Early asymptomatic or echo targeted:
This involves treating babies >12 hours of life based on echo findings of an unrestrictive PDA. There is some evidence that this method leads to a reduction in development of symptomatic PDA and pulmonary haemorrhage and exposes fewer babies to treatment than a prophylactic approach (8). This has the advantage of optimising treatment efficacy but this may still result in some over exposure to treatment compared with a symptomatic approach.
Early & Late Symptomatic Treatment:
Early symptomatic treatment selectively treats babies who have established signs of cardiac failure and pulmonary overcirculation on day 2-7 of life. Late symptomatic treatment selectively treats babies with established signs of cardiac failure and pulmonary overcirculation on day 10-14 of life. The presence of a large DA with cardiac structural normality should be confirmed on echo.
Early studies indicate that early treatment of the DA leads to reduced rates of chronic lung disease and reduced duration of mechanical ventilation when compared to late treatment (17).
Recent case series and studies excluding babies with the most symptomatic PDA suggest that moderately delayed PDA closure, with modest fluid restriction, can be well-tolerated and significantly reduce the need for medical treatment and/ or PDA ligation. However, there may be an increased rate of chronic lung disease and death (24).
Therefore, whilst a modestly delayed treatment approach may be tolerated by many babies, the impact on long-term outcome in the most fragile babies with early symptomatic PDA is unknown. Furthermore, although treatment at this stage is well tolerated, effectiveness of treatment on DA closure is reduced. This is because baby’s vasoconstrictive response is less prostaglandin-dependent and more dependent on other vasodilators (9).
Conservative or Supportive Treatment:
There is evidence that supportive management for PDA using newer modes of non invasive respiratory support, diuretics and fluid restriction can be tolerated by some babies and may reduce the need for medical treatment. However it is not clear if this approach is superior to medical closure. Animal models suggest long-term exposure to a left-to-right shunt is detrimental to lung mechanics and can lead to pulmonary hypertension (10) and the effects of a prolonged exposure to abnormal flow patterns on the cerebral circulation are unknown. In addition, fluid restriction can lead to nutritional deficits in preterm babies already reliant on high calorie intake to grow. This approach hasn’t been studied systematically. This may be well tolerated by some infants but it is important all infants being discharged home with a persistent PDA are referred to cardiology for follow up; either centrally or at a local outreach cardiology clinic.
Although generally well tolerated PDA ligation should not be the primary treatment of choice (15) and should be preceded by pharmacological treatment where possible. Ligation is not generally recommended in the first 3 weeks of life because of increased likelihood of adverse events. The optimal timing is unclear but for some babies, in significant cardiac failure, it can improve outcomes. Care should be taken over babies requiring prolonged periods of non invasive respiratory support where there can be significant left ventricular loading and potential mitral regurgitation and valve damage. If in any doubt seek a cardiology review or refer for assessment. If respiratory problems are predominant consideration of steroids and diuretics should be given prior to PDA ligation. Indications include:
- Failure to extubate
- Increasing oxygen requirement in association with an unrestrictive PDA
- Cardiac failure not controlled medically (CPAP dependency, failure to thrive)
- Feed intolerance or NEC (non acute)
- Compromised renal or cerebral blood flow + any of above
- Cardiology assessment that LV or mitral valve may be compromised.
Referral process
Patients should be referred to the ‘receiving’ Neonatal Consultant at the Royal Hospital for Children, Glasgow who will liaise with cardiac and cardiac surgical services.
SUMMARY 2- Pharmacological treatment should be considered where there is clinical and echocardiographic evidence of a moderate or large ductal shunt. - Pharmacological treatment should only be instigated when there is echocardiographic evidence of a significant DA.- Treatment duration should be determined by evidence of closure on echo. - Current medical therapies have poor efficacy beyond the early treatment period- Consider paracetamol as a 2nd line treatment.- Surgical ligation should be considered if there is poor response or contraindication to pharmacological treatment in the presence of cardiac failure and persistent requirement for respiratory support and/or systemic disturbance (feed intolerance, renal impairment, failure to thrive). - Cardiology review is important for babies with persisting evidence of PDA and cardiac failure, including those not requiring invasive ventilation.
Link to WoS Ibuprofen monograph here
Review by echo is recommended after the 3rd dose. If the Ductus Arteriosus remains patent a second course may be given 48 hours after the 1st course has completed.
Alternatively the initial course may be extended to 4 or 5 doses.
NB: There is evidence of increased efficacy with oral ibuprofen and the enteral route should be used where possible (>120mls/kg/day enteral feeds). Ibuprofen may be changed from IV to oral mid-course if feeds of 120ml/kg/day are reached.
Cautions: The immature kidney is very sensitive to the effects of PHGH2S inhibitors working on the cyclooxygenase or COX pathways (indomethacin and ibuprofen). The effects on urine output appear to be greatest in the beginning of the treatment course and rarely have clinical impact. Early treatment, in the first postnatal week, has been associated with spontaneous intestinal perforation. The association may be spurious but concurrent administration with corticosteroids should be avoided. Renal function and urine output should be monitored during treatment.
(NB at the time of writing Indomethacin has limited availability)
Link to WoS indomethacin monographs for prophylactic, early and late courses here
Review by echo is recommended after the 4th dose and courses shortened where possible (see monograph)
Cautions: The immature kidney is very sensitive to the effects of PHGH2S inhibitors working on the cyclooxygenase or COX pathways (indomethacin and ibuprofen). The effects on urine output appear to be greatest early in the treatment course but rarely have clinical impact. Effects begin to wane after several doses have been given. Early treatment, in the first postnatal week, has been associated with spontaneous intestinal perforation. The association may be spurious but concurrent administration with corticosteroids should be avoided. A prolonged course has been associated with increased rates of NEC (28) so dosing should always be titrated against echo assessment of constriction. Renal function and urine output should be monitored during treatment.
Link to WoS paracetamol monograph here
Both intravenous and oral preparations are available. Although toxicity in infants is rare (14) drug levels are required to optimise treatment and avoid toxicity. Careful attention should be paid to the monograph.
N.B: Paracetamol should not be changed between IV and oral mid-course without prior discussion with pharmacy.
Cautions: LFTs should be monitored if concerned about hepatic impairment.
- Evans NJ, Archer LNJ. Postnatal circulatory adaptation in term and healthy preterm newborns. Arch Dis Ch 1990;65:24e6.
- Clyman RI, Marayanan M. Patent ductus arteriosus: a physiological basis for current treatment practices. In: Current Topics in Neonatology. 4th edn. Hansen, TN: McIntosh N. W.B. Saunders Company, 2000.
- Kluckow M, Evans NJ. Ductal shunting, high pulmonary blood flow, and pulmonary hemorrhage. J Pediatr 2000;137:68e72.
- Noori S, McCoy M, Friedlich P, Bright B, Gottipati V, Seri I, et al. Failure of ductus arteriosus closure is associated with increased mortality in preterm infants. Pediatrics 2009;123:e138e44.
- Clyman RI, Ballard PL, Sniderman S, et al. Prenatal administration of betamethasone for prevention of patient ductus arteriosus. J Pediatr 1981;98:123–6.
- Benitz WE. Patent ductus arteriosus: to treat or not to treat? Arch Dis Ch Fetal Neonatal Ed 2012;97:F80e2.
- Fowlie PW, Davis PG, McGuire W. Prophylactic intravenous indomethacin for preventing mortality and morbidity in preterm infants. Cochrane Database of Systematic Reviews 2010;7:CD000174.
- Kluckow M, Jeffery M, Gill A, et al. A randomised placebo-controlled trial of early treatment of the patent ductus arteriosus. Arch Dis Ch Fetal Neonatal Ed. 2014;99:F99–104.
- Heuchan AM, Clyman RI. Managing the patent ductus arteriosus: current treatment options. Arch Dis Ch Fetal Neonatal Ed fetalneonatal-2014-306176 doi:10.1136/archdischild-2014-306176
- McCurnin D, Seidner S, Chang LY, et al. Ibuprofen-induced patent ductus arteriosus closure: physiologic, histologic, and biochemical effects on the premature lung. Pediatrics 2008;121:945–56.
- Heuchan AM, Young D. Early colour Doppler duct diameter and symptomatic patent ductus arteriosus in a cyclo-oxygenase inhibitor naïve population. Acta Paediatr 2013;102:254–7.
- Ohlsson A, Walia R, Shah SS. Ibuprofen for the treatment of patent ductus arteriosus in preterm and/or low birth weight infants. Cochrane Database of Systematic Reviews 2010, Issue 4. Art. No.: CD003481. DOI: 10.1002/14651858.CD003481.pub4.
- Oncel, M. Y., Yurttutan, S., Erdeve, O., Uras, N., Altug, N., Oguz, S. S., ... & Dilmen, U. (2014). Oral paracetamol versus oral ibuprofen in the management of patent ductus arteriosus in preterm infants: a randomized controlled trial. The Journal of pediatrics, 164(3), 510-514.
- Penna A ,Buchanan N 1991 Paracetamol poisoning in children and hepatotoxicity. Br J Clin Pharmacol 32:143-149
- Heuchan AM, Hunter L, Young D. Outcomes following the surgical ligation of the patent ductus arteriosus in premature infants in Scotland. Arch Dis Child Fetal Neonatal 2012;97:F39-44. doi:10.1136/adc.2010.206052
- Cotton RB, Stahlman MT, Kovar I, Catterton WZ. Medical management of small preterm infants with symptomatic patient ductus arteriosus. The Journal of pediatrics. 1978 Mar 31;92(3):467-73.
- Clyman RI. Recommendations for the postnatal use of indomethacin: an analysis of four separate treatment strategies. J Pediatr 1996;128:601–7.
- Smith A, Maguire M, Livingstone V, Dempsey EM. Peak systolic to end diastolic flow velocity ratio is associated with ductal patency in infants below 32 weeks of gestation. Arch Dis Child Fetal Neonatal 2015; Mar 1;100(2):F132-6.
- Ohlsson A, Walia R, Shah SS. Ibuprofen for the treatment of patent ductus arteriosus in preterm and/or low birth weight infants. Cochrane Database Syst Rev 2013;4:CD003481.
- Dani C, Vangi V, Bertini G, et al. High-dose ibuprofen for patent ductus arteriosus in extremely preterm infants: a randomized controlled study. Clin Pharmacol Ther 2012;91:590–6.
- Desfrere L, Zohar S, Morville P, et al. Dose-finding study of ibuprofen in patent ductus arteriosus using the continual reassessment method. J Clin Pharm Ther 2005;30:121–32.
- McNamara PJ, Sehgal A. Towards rational management of the ductus arteriosus – the need for staging. Arch Dis Child Fetal Neonatal Ed. 2008 Jan; 93(1):F78.
- Neumann R, Schulzke SM, Buhrer C. Oral ibuprofen versus intravenous ibuprofen or intravenous indomethacin for the treatment of patent ductus arteriosus in preterm infants: a systematic review and meta-analysis. Neonatology 2012;102:9–15.
- Kaempf JW, Wu YX, Kaempf AJ, et al. What happens when the patent ductus arteriosus is treated less aggressively in very low birth weight infants? J Perinatol 2012;32:344–8.
- Garland J, Buck R, Weinberg M. Pulmonary hemorrhage risk in infants with a clinically diagnosed patent ductus arteriosus: a retrospective cohort study. Pediatrics. 1994;94:719–23.
- Alfaleh, K., Smyth, J. A., Roberts, R. S et al (2008). Prevention and 18-month outcomes of serious pulmonary hemorrhage in extremely low birth weight infants: results from the trial of indomethacin prophylaxis in preterms. Pediatrics, 121(2), e233-e238.
- Gersony WM, Peckham GJ, Ellison RC, Miettinen OS, Nadas AS. Effects of indomethacin in premature infants with patent ductus arteriosus: results of a national collaborative study. J Pediatr. 1983 Jun;102(6):895-906.
- Herrera CM, Holberton JR, Davis PG. Prolonged versus short course of indomethacin for the treatment of patent ductus arteriosus in preterm infants. The Cochrane Library. 2007 Jan 1.
Last reviewed: 22 May 2017
Next review: 31 December 2023
Author(s): Dr Andrew MacLaren – Neonatal Grid Trainee, Royal Hospital for Children, Glasgow; Dr Anne Marie Heuchan – Neonatal Consultant, Royal Hospital for Children, Glasgow
Co-Author(s): Other Professionals Consulted: Peter Mulholland – Neonatal Pharmacist, Royal Hospital for Children, Glasgow
Approved By: West of Scotland Neonatology Managed Clinical Network

