Congenital hypothyroidism in Scotland, guidelines for the management of
exp date isn't null, but text field is
Objectives
- To guide the management of infants referred with an elevated TSH concentration on Guthrie blood spot screening.
- To give guidance on the initial investigations for infants diagnosed with congenital hypothyroidism.
- To give guidance for ongoing management and monitoring of infants and children with congenital hypothyroidism.
Scope
Patients to whom this document applies:
- Infants in Scotland with an elevated TSH on Guthrie blood spot screening.
Audience
Who should use this document?
- General paediatricians
- Neonatologists
- Paediatricians with an interest in paediatric endocrinology
- Paediatric endocrinologists.
Diagnosis:
- A screening test is performed on a newborn blood spot sample for the measurement of thyroid stimulating hormone (TSH). A high TSH suggests hypothyroidism.
- Confirmatory tests:
- pre-treatment thyroid function tests (TSH and free thyroxine – fT4)
- quantitative thyroglobulin (Tg).
- Optional confirmatory tests may include:
- thyroid imaging (ultrasound and/or radio-isotope scans of the neck, sub-lingual and upper chest area)
- thyroid autoantibodies (thyroid peroxidase and TSH receptor antibodies) in the infant (and if indicated, mother)
- maternal TFTs
- Genetic testing
Treatment:
Replacement therapy with levothyroxine (thyroxine, L-thyroxine, LT4).
- To bring thyroid function into the normal range as rapidly as possible while avoiding adverse effects.
- To maintain fT4 in the upper quartile of the normal reference range and concurrent TSH in the normal range.
- To diagnose the cause of congenital hypothyroidism following positive screening.
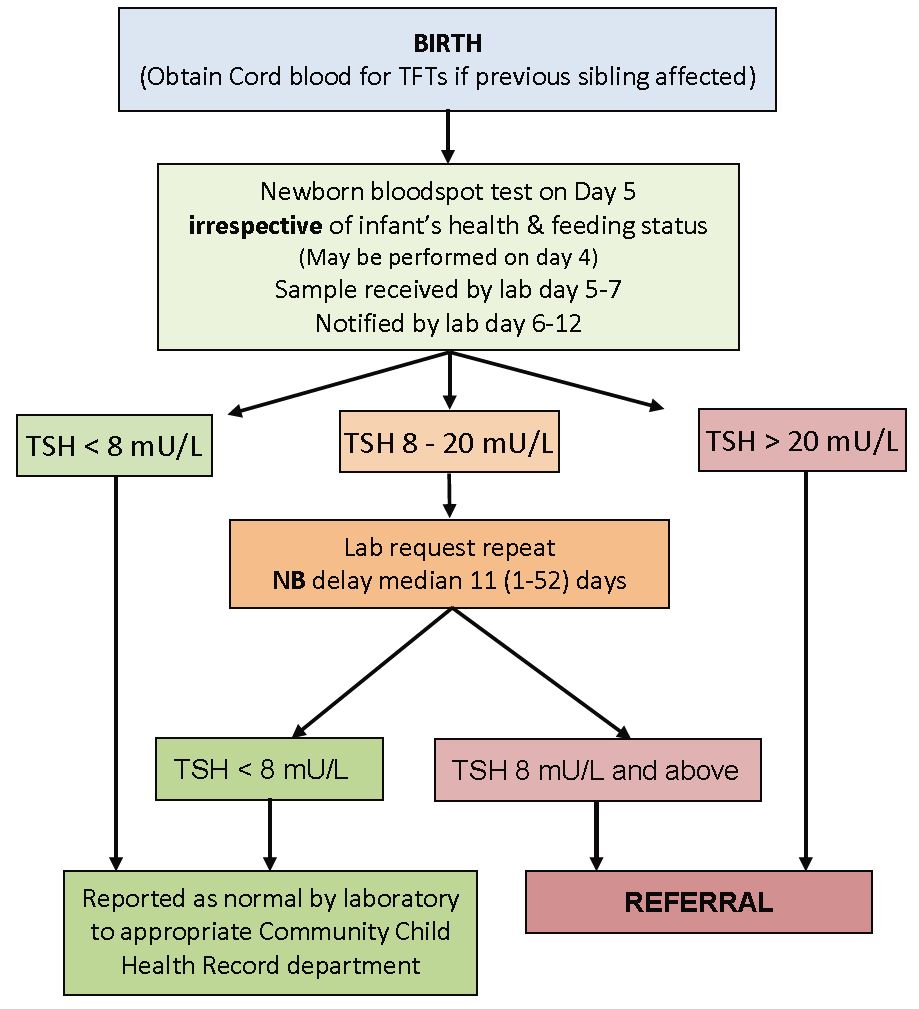
Referral procedure by laboratory:
Laboratory staff notifying abnormal result must speak directly to a Consultant Paediatrician – either one of the two named paediatricians shown in the table below or the on-call paediatrician for the maternity unit.
NB all results are notified simultaneously to the person managing the Scottish CHT Register at RHC Glasgow.
Table 1 – Named paediatricians for Notification of Raised TSH
|
Centre |
1st named paediatrician |
2nd named paediatrician |
Default |
|
Ayrshire |
Scott Williamson |
Jon Staines |
On-call paediatrician in maternity/ neonatal unit |
|
Borders |
Graeme Eunson |
Andy Duncan |
|
|
Dumfries |
Rajendran Shyam |
On Call consultant |
|
|
Fife |
Anthony Tasker |
Jo Watt |
|
|
Forth Valley |
John Schulga |
Michael Colvin |
|
|
Glasgow |
Guftar Shaikh |
Avril Mason |
|
|
Grampian |
Amalia Mayo |
Craig Oxley |
|
|
Highland |
Stuart Henderson |
Alan Webb |
|
|
Lanarkshire |
Iain Hunter |
Shubhangi Shewale |
|
|
Lothian |
Via Paed endo secretary |
Endocrine Cons. of week |
|
|
Paisley |
Graham Stewart |
Hilary Conetta |
|
|
Tayside |
Nicky Conway |
Clare Webster |
Table 2 - Telephone numbers
|
Centre |
Hospital |
Switchboard |
|
Ayrshire |
Crosshouse, Ayr |
01563 521133 |
|
Borders |
Borders General |
01896 826000 |
|
Clyde |
Royal Alexander, Paisley |
0141 887 9111 |
|
Dumfries |
Dumfries General |
01387 246246 |
|
Fife |
Victoria Hospital |
01592 643355 |
|
Forth Valley |
Forth Valley Royal, Larbert |
01324 566000 |
|
Glasgow |
Royal Hospital for Children, Glasgow |
0141 201 0000 |
|
Glasgow |
Princess Royal Maternity Hospital |
0141 211 5400 |
|
Grampian |
Children’s Hospital, Aberdeen |
0845 456 6000 |
|
Highland |
Raigmore, Inverness |
01463 704000 |
|
Lanarkshire |
Wishaw General, Wishaw |
01698 361100 |
|
Lothian |
Royal Hospital for Sick Children, Edinburgh |
0131 536 0611 |
|
Lothian |
Royal Infirmary, Edinburgh |
0130 536 1000 |
|
Tayside |
Ninewells, Dundee |
01382 660111 |
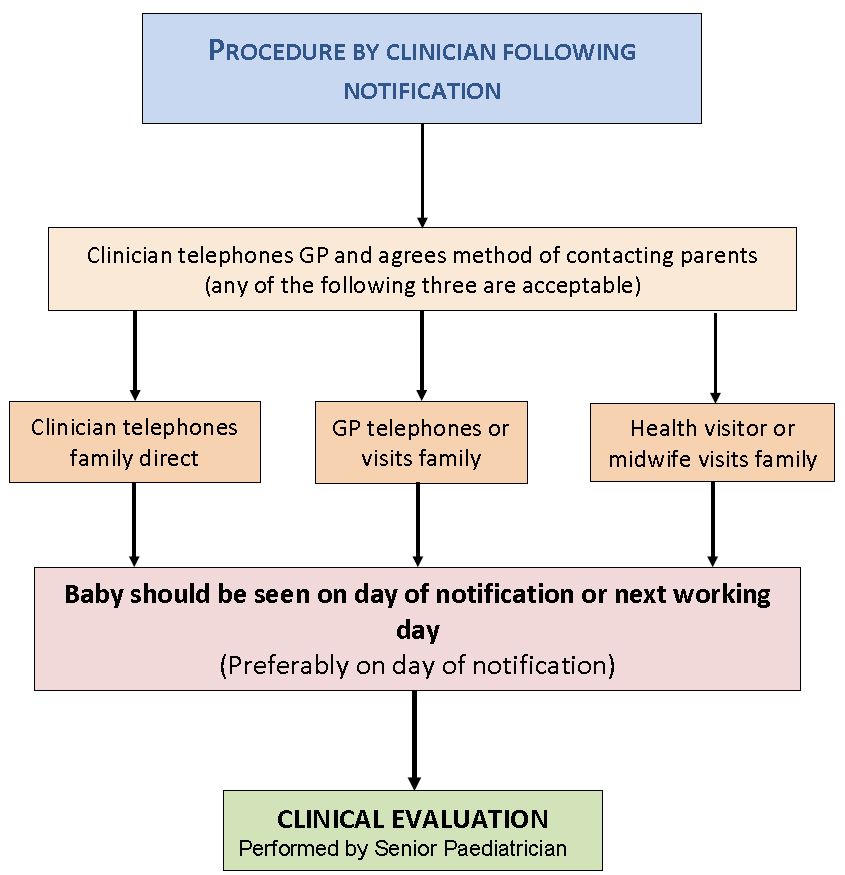
HISTORY – to include:
-
Whether a sibling is affected.
-
A family history of thyroid illness/problems.
-
A history of thyroid disease or anti-thyroid therapy in the mother.
-
Any symptoms of hypothyroidism (e.g. poor feeding, sleepiness, jaundice, constipation, cold peripheries, hoarse cry).
EXAMINATION – to include:
-
Measurement of weight, head circumference and length.
-
Measurement of (c.f. reported) parental heights.
-
Presence or absence of goitre.
-
Signs of hypothyroidism (e.g. coarse facies, hoarse cry, umbilical hernia).
INITIAL INVESTIGATION:
-
A minimum of 1 mL of blood for TFTs in heparinised bottle or paediatric heparin tube (be prepared to make several attempts in order to get sufficient blood). (It is vital that sufficient blood is taken)
-
1 mL clotted blood for quantitative thyroglobulin.
-
Blood for genetics if sufficient blood is available (sample can be stored for use later).
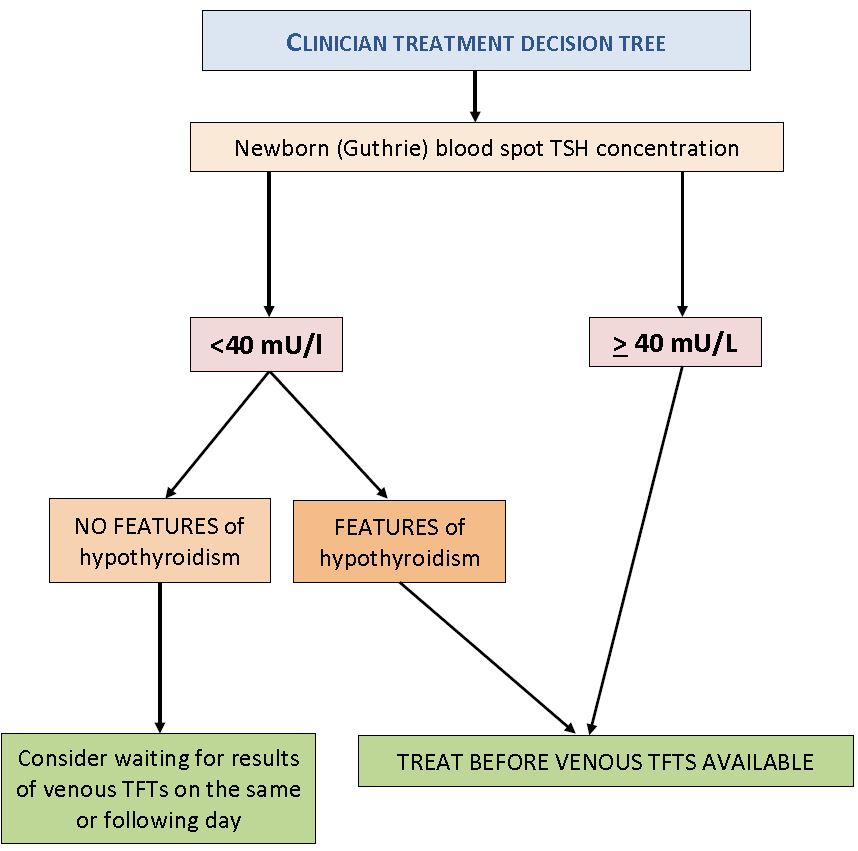
This decision tree is based on referenced evidence (1,2)
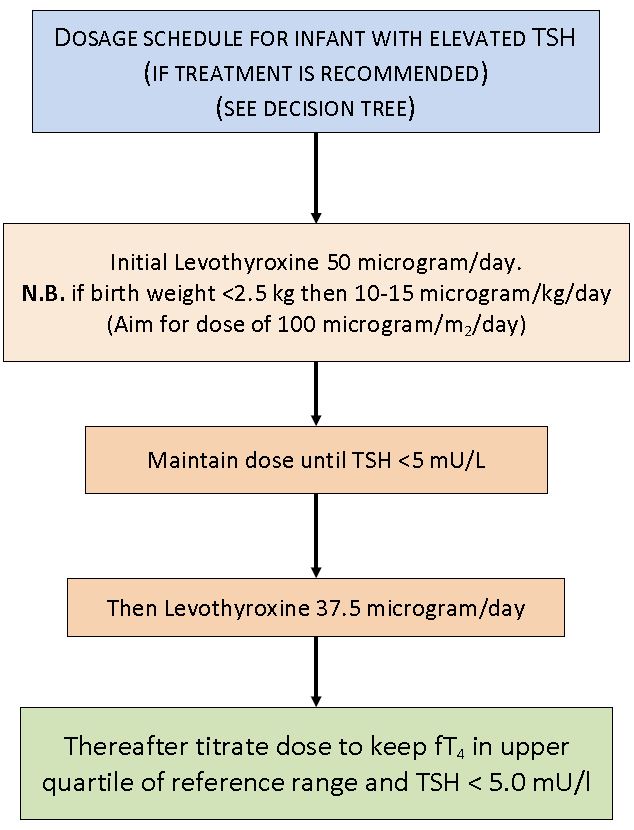
Written instructions on how to give thyroxine should be available and supplied to the parents.
LevoThyroxine Tablets
- Available as 12.5 microgram, 25 microgram, 50 microgram, 75 microgram or 100 microgram strength.
- The tablets should be crushed and mixed with a small volume (less than 5ml) of liquid (formula, expressed breast milk or boiled and cooled water).
- If necessary, a tablet cutter for halving tablets should be supplied to the family.
- The first dose should be given by parents under the supervision of a nurse or pharmacist.
SPEG does not recommend using any of the liquid forms of thyroxine which are available including syrups, solutions and suspensionsTablets have been used extensively and successfully in the management of congenital hypothyroidism and therefore SPEG recommends the use of tablets, in accordance with European Society for Paediatric Endocrinology guidelines (ESPE) [1]. Tablets are also much cheaper to use than other preparations.
ESSENTIAL INFORMATION TO GIVE AT THE INITIAL VISIT:
- Thyroid hormone is vital for normal brain development and somatic growth
- The thyroid gland is either absent, too small or not producing thyroid hormone properly
- It is likely to be permanent, or there is insufficient evidence to determine whether or not it is permanent
- With treatment, it has a good prognosis.
- Adherence to treatment is crucial, especially from birth to 3 years of age when the brain is developing most rapidly
- The treatment is easy to administer, but should be given with care.
- Verbal and written instructions should be given on how to give medicines.
It is important to give the family written information about congenital hypothyroidism.
There are several excellent leaflets and booklets available (National Newborn screening, British Society for Paediatric Endocrinology and Diabetes (BSPED)):
Congenital hypothyroidism (CHT) suspected: description in brief (Public Health England, last updated April 2018)
Congenital hypothyroidism (CHT) confirmed: description in brief (Public Health England, last updated April 2018)
Hypothyroidism (underactive thyroid gland) in childhood (BSPED)
How to give thyroxine to babies and children (SPEG, last updated August 2017)
ESPE consensus statement (1) on Congenital Hypothyroidism recommends thyroid imaging for all babies with suspected congenital hypothyroidism because:
- It is more informative than blood tests alone
- It will aid in guiding options for genetic counselling
- With the results, it is possible to determine the likelihood of lifelong treatment if permanent Congenital Hypothyroidism is proven
- It provides a useful additional guide as to thyroxine dose (agenesis is likely to require larger doses).
Isotope scans should be performed by day 5 of the start of treatment[5] to ensure the avoidance of false negatives, due to TSH suppression (it is advisable to check thyroid function on the day of scans to confirm the reliability of results).
Currently, if imaging is to be performed, the Hospital for Children, Glasgow is currently the only site that is able to perform these scans. If scanning is to be considered, there should be early discussion with a paediatric endocrinologist.
- Imaging should never be allowed to delay the initiation of treatment.
If thyroid imaging suggests that the cause of the hypothyroidism might be due to an enzyme defect in the function of the thyroid gland, there is a mutation analysis service available in Scotland. This is based in the genetics laboratories at the Queen Elizabeth University Hospital.
The service offers analysis for the three most common mutations responsible for dyshormonogenesis in Scotland, namely mutations in thyroperoxidase (TPO), thyroglobulin (Tg) and the TSH receptor (TSHR) genes.
For genetic testing, a sample of blood in an EDTA bottled is required. The lab requires at least 1 ml volume.
Samples should be obtained from the infant, and also from both parents. This can be performed at a subsequent follow-up visit as it may be difficult to obtain sufficient blood samples at the initial diagnostic visit.
The service is managed by Dr Therese Bradley who can be contacted on: therese.bradley@ggc.scot.nhs.uk or by telephone on 0141 354 9330
If unavailable the duty scientist can be contacted on: geneticlabs@ggc.scot.nhs.uk

N .B We would also suggest additional clinic visits following dosage alteration or when there are problems with poor adherence.
At puberty/rapid growth or weight gain, more frequent monitoring is recommended
-
Calculate the optimal dose and adjust dosage pre-emptively, using Thyroid Function Tests to confirm adherence.
-
Assess Growth:
- weight measurement
- supine length measurement until 2 years, then height measurement
- head circumference measurement until 3 years
-
Developmental assessment:
- Consider pre-school formal audiology (for subtle hearing impairment due to Congenital Hypothyroidism [1])
- if aged <4-5 years, developmental progress
- aged over 4-5 years, school progress
- Consider neurodevelopmental assessment.
- Consider pre-school formal audiology (for subtle hearing impairment due to Congenital Hypothyroidism [1])
-
Education:
- from secondary school onwards ensure that patient has reasonable knowledge of CH.
-
Adult transfer:
- Boys – to GP.
- Girls – to GP, give pre-pregnancy counselling.
Transfer to Primary care for 6 to 12 monthly monitoring of TFT’s
Aim for: fT4 in the upper quartile of reference range and TSH <5.0 mU/l (but avoid undetectable TSH)
This may be required if no cause has been identified for the hypothyroidism in the neonatal period and should be considered after the age of 3 years.
Re-evaluation should be considered in patients with a normally sited thyroid gland and/or those who have had no increase in thyroxine requirement since infancy.
This can consist of either:
- An initial reduction in thyroxine dose followed by monitoring of thyroid function and further reductions in thyroxine dose and monitoring of thyroid function if results remain normal, until thyroxine is discontinued.
- Discontinuation of thyroxine with thyroid function assessment in 4-6 weeks.
If the TSH is elevated, hypothyroidism is confirmed and treatment should be recommenced as soon as possible.
Re- evaluation is not indicated if there has been a rise in the TSH in the first year, if there is a genetic cause or if there is an abnormal gland on imaging.
- Leger J, Olivieri A, Donaldson M, Torresani T, Krude H, van VG, Polak M, Butler G: European Society for Paediatric Endocrinology consensus guidelines on screening, diagnosis, and management of congenital hypothyroidism. Horm Res Paediatr 2014;81:80-103.
- Pokrovska T, Jones J, Shaikh MG, Smith S, Donaldson MD: How well does the capillary thyroid-stimulating hormone test for newborn thyroid screening predict the venous free thyroxine level? Arch Dis Child 2016;101:539-545.
- Jones JH, Gellen B, Paterson WF, Beaton S and Donaldson MDC. Effect of high versus low initial doses of L-thyroxine for congenital hypothyroidism on thyroid function and somatic growth. Arch Dis Child (2008); 93: 940-44.
- Selva KA, Harper A, Downs A, Blasco PA and LaFranchi SH. Neurodevelopmental outcomes in congenital hypothyroidism: comparison of initial T4 dose and time to reach target T4 and TSH. J Pediatr 2005; 147: 775-780.
- Perry RJ, Maroo S, Maclennan AC, Jones JH and Donaldson MDC. Combined ultrasound and isotope scanning is more informative in the diagnosis of congenital hypothyroidism than single scanning. Arch Dis Child 2006; 91: 972-976.
Last reviewed: 24 May 2022
Next review: 24 May 2023
Author(s): Jeremy Jones; M Guftar Shaikh; John Schulga
Version: 1.1
Co-Author(s): Group Committee: SPEG NMCN Steering Group; SPEG Guidelines sub-group
Approved By: SPEG Guidelines Group and West of Scotland Neonatal MCN Guideline Group
Document Id: 383

