Policy and Guidelines for the Safe Prescribing, Dispensing and Administration of Systemic Anti-Cancer Therapy (SACT) for Children, Teenagers and Young Adults under the Care of the Haematology/Oncology Team RHC, Glasgow (Schiehallion Ward and Schiehallion Daycare Unit)
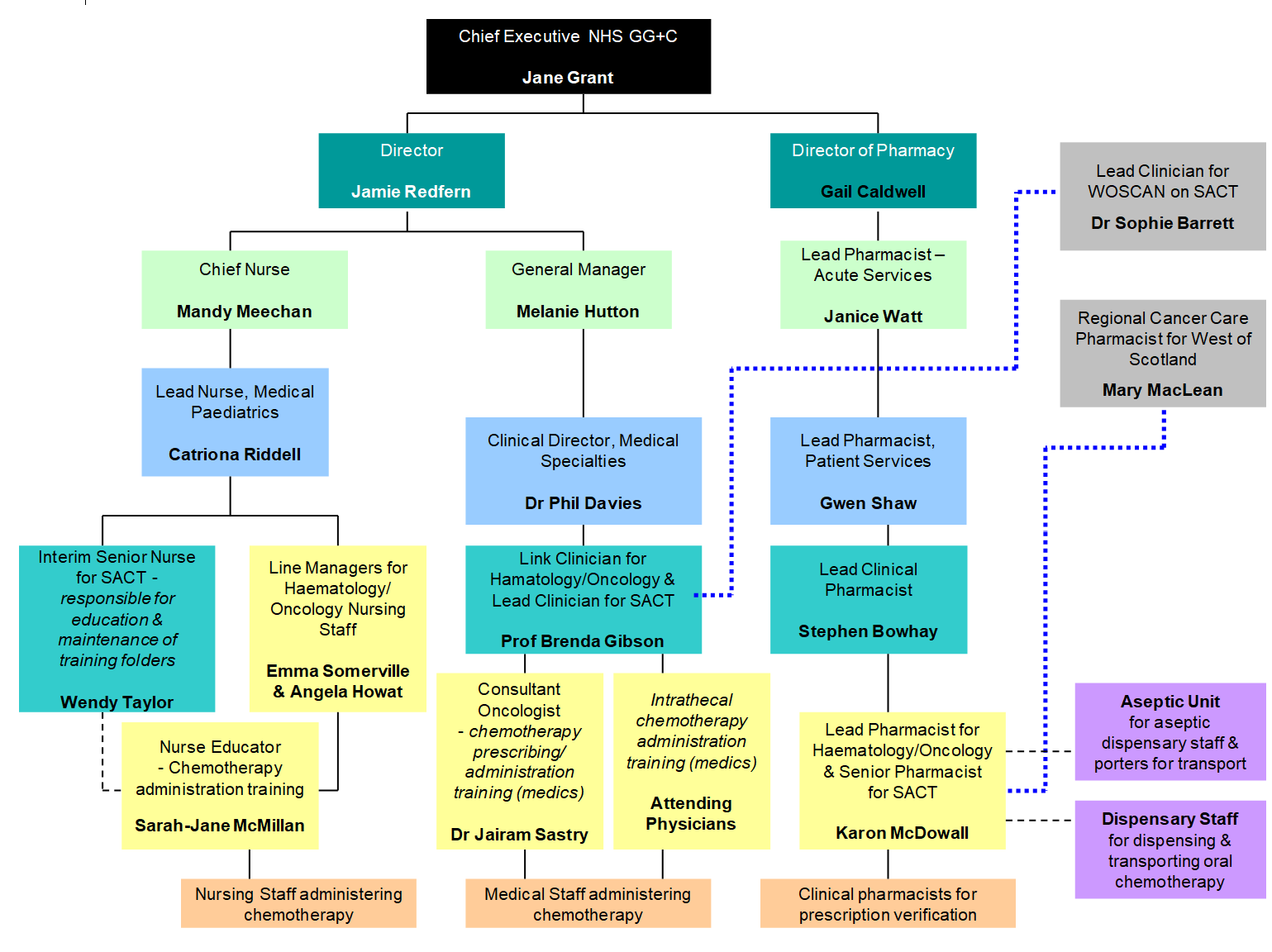
The overall responsibility for the safe delivery of systemic anti-cancer therapy (SACT) rests with the Chief Executive of Greater Glasgow and Clyde. This responsibility is delegated to the SACT Lead for GG+C (Sophie Barrett) who then further delegates this to Professor Brenda Gibson (Haematology Consultant & Link Clinician for Haematology/Oncology) as Lead Clinician for SACT for paediatric services – see Appendix 1 - Reporting Structure
Systemic anti-cancer therapy (SACT) encompasses both cytotoxic chemotherapy and biological therapies. Cytotoxic chemotherapies are potentially carcinogenic, mutagenic and hazardous (as defined by the Control of Substances Hazardous to Health Regulations 2002- COSHH). The risks to patients receiving cytotoxic chemotherapy are well documented and must be balanced against clinical benefit. The risk to staff through occupational exposure is less clear. However, there is sufficient evidence to support the implementation of all necessary measures to limit exposure.
SACT must be prescribed, dispensed, administered and disposed of in accordance with the Medicines Act 1968. CEL 30 (2012), Guidance on the safe use of SACT, describes both best practice and the clinical governance framework for all services delivering SACT in NHS Scotland.
This Standard Operating Procedure (SOP) should be read alongside other relevant and related policies.
These guidelines refer to practice in relation to SACT administration (by all routes), when used for the treatment of cancer.
The term Chemotherapy Handling refers to all elements/pathways of chemotherapy delivery from prescription, dispensing, administration and disposal of (Appendix 2).
The term Practitioner refers to the healthcare professional undertaking prescribing, dispensing and/or administration of chemotherapy. A practitioner is a qualified and appropriately trained nurse, doctor, pharmacist or pharmacy technician. These practitioners must work within their professional competencies.
The term carer refers to the parent /carer who administers oral SACT and to the health care professional or parent/carer who may be dealing with patient waste. This will include any person who may be in contact/handling urine, faeces or vomit from a patient who is receiving SACT.
These guidelines assume that local practices and policies for education and training are in place, that adherence is regularly monitored and audited, and that practices and policies are updated.
2.1 Procedure for Performing a Diagnostic Lumbar Puncture & for the Administration of Intrathecal Chemotherapy (RHC-HAEM-ONC-012)
2.2 Administration of Intrathecal Chemotherapy (RHC-HAEM-ONC-013)
2.3 Intrathecal SACT Assessment Questions (RHC-HAEM-ONC-015)
2.4 Spillage Procedures for Chemotherapy (RHC-HAEM-ONC-005)
2.5 Prevention, Treatment & Follow-up in the Event of Suspected Cytotoxic Extravasation (RHC-HAEM-ONC-009)
2.6 NHSGGC Vascular Access Procedure and Practice Guideline - hospital policy via Staffnet
2.7 Infection Control policy – hospital policy via NHSGGC website
2.8 Needlestick & Exposure to Bodily Fluids policy – hospital policy via Staffnet
2.9 Safe & Secure Handling of Medicines policy – hospital policy via Staffnet
2.10 Control of Substances Hazardous to Health (COSHH) regulations – hospital policy via Staffnet
2.11 Resuscitation guidelines –Resuscitation page on Staffnet
2.12 GG&C Policy for the Out of Hours Supply of SACT – hospital policy via Staffnet
2.13 Pharmacy Staff Competencies Training Record (RHC-HAEM-ONC 052)
Departmental SOPs can be accessed electronically via Q-Pulse. Contingency hard copies are also retained within the white lever arch files in the Dr’s office on the Schiehallion Ward and Schiehallion Daycare Unit. Hospital policies are accessed via the Clinical Guidelines section of Staffnet.
The following staff must be aware of this policy and understand its impact on the delivery of SACT:
- Medical
- Nursing
- Pharmacy
- Administration staff
- General services staff
All staff are responsible for documenting clinical incidents relating to SACT on Datix as soon as possible after the incident has occurred or been discovered.
Lead Roles for Safe Delivery of Systemic Anti-Cancer Therapy (SACT)
3.1 Lead Clinician – Professor Brenda Gibson
- Accountable for the safe delivery of SACT which includes :
- Establishing systems to ensure compliance with current legislation, national standards and guidelines.
- Ensuring systems are in place to develop, approve, implement and regularly review policies, protocols and guidelines for the safe delivery of SACT, including document control. Complying with a cross regional audit programme with external peer review from another cancer network to provide assurance on the safe delivery of SACT.
- Ensuring non-conformance remediation plans, with priorities & timescales assigned, are in place to achieve full compliance with CEL 30 (2012) standards.
- Ensuring that there is a programme of education and training for all staff involved in the delivery of SACT services.
The following staff support the Lead Clinician in the safe delivery of SACT in their specific areas:
3.2 Pharmacy support
- Karon McDowall – Lead Pharmacist for Paediatric Haematology and Oncology and pharmacy lead for SACT delivery
- Carol Mitchell – Senior Cancer Care Pharmacist with specific responsibilities for the delivery of pharmaceutical care to haematology/oncology inpatients, haemopoietic stem cell transplant patients and patients attending the leukaemia clinic. Additional responsibilities include the clinical pharmacy training of band 6 pharmacists
- Lorna Wilson – Senior Cancer Care Pharmacist with specific responsibilities for chemotherapy electronic prescribing and palliative care.
- Band 7 (1.6wte) pharmacists from the main paediatric rotation and 1 wte specialist band 7 for electronic prescribing and clinical pharmacy input to the DCU.
3.3 Nursing Support
- Ward Managers (Emma Somerville & Kirsteen Meikle - Inpatient Ward & Angela Howat - Daycare Unit)
- Validation of ChemoCare documentation
- Advanced Nurse Practitioner
- Maintenance of Training Folders (both paper & electronic) for medical and nursing staff
- Sarah-Jane McMillan - Nurse Educator
- Training & Education of Nursing Staff on Administration & Safe Handling of SACT and Management of Extravasation & Spillages
- Paediatric Oncology Outreach Nurses (POONs)
- Safe delivery of SACT outwith the cancer centre in patient’s home
3.4 Medical Support
- Dr Jairam Sastry - Consultant Oncologist
- Training & Education of Medical Staff
- Consultant Haematologists / Oncologists
- Validation of ChemoCare documentation
3.5 Administrative Support
- Quality Manager
- Document control of protocols and template prescriptions provided by senior pharmacist and data managers
- Document control of supportive care guidelines
- Administrative support for audit activity
- Data Managers
- Maintenance of patient shadow notes which hold essential documentation required for treatment delivery. These may be used for inspections and audit activity.
- Maintain comprehensive documentation related to trial and non trial activity
4.1 Information and Support given to Parents/Patients by Pharmacy
Both verbal and written information must be available for parents/patients. Clear explanation and instruction must be given throughout the administration of anti-cancer agents. This should include both general and specific side-effects of the drug(s) being given.
- Individual patient information leaflets on the administration of oral chemotherapy at home are available for parents/patients from Pharmacists based within the Haematology/Oncology Unit. These include the anticipated benefits, potential risks, toxicities and information on safe handling & disposal.
- Tablets (or liquid for children who cannot swallow tablets) will be given to patients/parents for home administration.
- Advice must be given on the safe handling of SACT.
- Parents are informed that they should bring back all home supplies of medicines at each admission or clinic visit, with the exception of the leukaemia clinic where patients are advised to return all home medication supplies on a monthly basis.
4.2 Areas Suitable for Intrathecal Chemotherapy Drug Administration (see individual door signage – if unsure please check with Ward Manager)
- Treatment room SCH-079 within the Schiehallion Inpatient Ward
- Treatment Room 1 within the Schiehallion Daycare Unit
- Areas where general anaesthesia is administered to the patient (ie Theatre suite, MRI, interventional radiology and PICU )
NB: UNDER NO CIRCUMSTANCES SHOULD INTRAVENOUS AND INTRATHECAL DRUGS BE ADMINISTERED AT THE SAME TIME OR IN THE SAME ROOM/AREA
PATIENTS WHO ATTEND DAY SURGERY FOR ADMINISTRATION OF INTRATHECAL THERAPY AND/OR BONE MARROW ASPIRATE, BUT THE PROCEDURE IS CANCELLED, MUST STILL NEVER RECEIVE INTRAVENOUS VINCA ALKALOIDS ON THAT DAY.
4.3 Areas Designated as Suitable for Intravenous Chemotherapy Administration (see individual door signage – if unsure please check with Ward Manager)
- Bedside within all Schiehallion Inpatient Ward cubicles
- Allocated treatment rooms within the Schiehallion Daycare Unit
- Outpatient department treatment room
- PICU chemotherapy may be given by all routes within the PICU. Irrespective of site of administration, all chemotherapy must be checked by two members of SACT trained staff
- In exceptional circumstances patients may receive chemotherapy treatment in other areas of the hospital. Again this must always be checked by two members of SACT trained staff and administered by an appropriately trained individual
NB: ADMINISTRATION OF INTRAVENOUS OR INTRATHECAL CHEMOTHERAPY MUST ONLY BE CARRIED OUT IN DESIGNATED AREAS
5.1 Training
All nurses who are new to the Schiehallion Inpatient Ward and Daycare Unit receive induction training which will include an overview of SACT. This will be the responsibility of the Clinical Nurse Educator.
Nursing staff who are involved in the administration of chemotherapy must undergo a Systemic Anti Cancer Therapy course prior to any independent involvement in any aspect of SACT administration.
Nursing staff new to oncology may be involved in the delivery of intramuscular and oral chemotherapy but only under the direct supervision of a chemotherapy trained nurse. All doses of SACT must be checked and signed for by two chemotherapy certified nurses who will check that the name on the patient’s wrist band matches the name on the printed prescription and on the drug product.
SACT must be prescribed on specific chemotherapy prescriptions. A SACT trained nurse in a nurse who has been passed as competent to deliver infusional chemotherapy. Nurses are only deemed competent to administer intravenous bolus chemotherapy when additional training has been completed.
All nursing staff must be aware of and understand the following:
- Local medicines policy
- Local intravenous flush policy
- Procedure for Performing a Diagnostic Lumbar Puncture & Administration of Intrathecal Chemotherapy (RHC-HAEM-ONC-012)
- Administration of Intrathecal Chemotherapy (RHC-HAEM-ONC-013)
- Intrathecal SACT Assessment Questions (RHC-HAEM-ONC-015)
- Spillage Procedures for Chemotherapy (RHC-HAEM-ONC-005)
- Vascular Access Procedure and Practice Guidelines (GG&C Clinical Guidelines Electronic Resource Directory)
- The working of mechanical pumps and other devices covered in induction training
- The signs and complications of myelosuppression
- The common SACT side effects including nausea, vomiting, stomatitis, diarrhoea, phlebitis and alopecia
- Local guidance on chemotherapy related oncological emergencies including extravasation, anaphylaxis and neutropenic sepsis
- The West of Scotland Cancer Network (WOSCaN) Extravasation Guideline and implications in practice
- CVAC competency and must be assessed by the Clinical Nurse Educator
5.1.1 Annual Updates:
Nurses involved in the delivery of SACT must complete the E-learning (Learn-Pro) module on chemotherapy / SACT delivery annually.
5.1.2 Biannual Updates:
Nurses involved in any aspect of the administration of intrathecal chemotherapy must complete the chemo competency IT assessment – RHC-HAEM-ONC-015 every 2 years.
Intrathecal cytotoxic drugs can only be checked by a registered nurse who is listed on the directorate intrathecal register for this role.
5.2 Pre-administration verification checks – prior to first and subsequent cycles of SACT
- Nurses must check that the patient/carer has been fully informed about the chemotherapy, that age-appropriate written information has been provided, that written consent has been taken for the proposed chemotherapy and that the procedure has been fully explained.
- The protocol and first cycle of SACT must be assigned by a consultant Oncologist/Haematologist. The treatment plan, protocol and flow sheets with the written consent form must be placed in the patient’s extended scanning folder.
- All documentation must have the patient’s addressograph label attached. Patients with only an electronic record must have a summary of their intended treatment recorded by their consultant on the relevant e-forms on Clinical Portal.
- The nurse must check that the prescription is dated correctly, signed, written clearly and unambiguously and is in accordance with the chemotherapy protocol. She/he must also check that the prescription has been authorised by a designated chemotherapy competent Oncology/Haematology doctor or advanced nurse practitioner and has been verified by pharmacy.
- She/he must check the body surface area and that dose calculations are in accordance with the protocol with particular reference to dose modifications due to age or weight - e.g. less than 6 months, less than 10/12 kgs, or management of underweight/overweight patients (BMI).
- She/he must check that all pre-chemotherapy investigations have been completed and results reviewed by an Oncology/Haematology doctor or advanced nurse practitioner. The results must be entered onto Chemocare or the SACT authorisation sheet.
- She/he must check that appropriate supportive care medications have been prescribed and administered.
- She/he must check that the name and CHI number on the patient’s name band corresponds with the name on the prescription chart. Both inpatients and day case patients must wear a name band. The patient’s name, date of birth and CHI number should be confirmed with the patient and/or carer.
- Chemotherapy doses must be administered on the date and at the time and interval stated on the prescription in accordance with the protocol.
- The name of the drug, the dose, the route of administration and, where applicable, the infusion fluid must be identical on both the prescription and pharmacy label.
- She/he must check the expiry date and timings of all drug doses and ensure that the expiry date will not be exceeded before the administration is complete.
- She/he must check all parenteral doses for particulate contamination e.g. precipitation before administration.
- A minimum of 30mls of compatible solution should be used to flush intravenous lines between intravenous drugs. Lines should be primed with the correct infusion fluid and the rate of the infusion prime should be equivalent to the drug infusion rate.
- Both the nurse administering, and the nurse checking, the chemotherapy must sign the appropriate sections of the administration document.
- If, for any reason (poor access / problem during administration), the dose, or part thereof, is not administered, the volume administered and/or reasons for discontinuation of treatment should be clearly documented. Schiehallion medical and pharmacy staff should be notified and part doses disposed of in designated sharps boxes in accordance to COSHH and waste management policy.
5.3 Venous Access
- An appropriate vascular access device should be placed which will meet the needs of the proposed treatment.
- Chemotherapy should NOT be given if there is any doubt about the patency of the venous access device.
- Venous access should be assessed prior to the first treatment and throughout subsequent cycles.
- Only nursing or medical staff who have been trained and assessed as competent for cannulation should cannulate a patient who is to receive chemotherapy.
- Ideally a new cannula should be placed for all peripheral vesicant bolus administration. If this is not possible then one that has been in place for less than 48 hours should be used. It must be patent and bleed back easily.
- Central venous access is the route of choice for drugs or fluids which are to be infused over a long period, are an irritant to the peripheral veins, or have the potential to cause tissue necrosis.
- Some therapies will justify the placement of non-tunnelled, percutaneous central venous catheters. However, several months of intensive therapy indicate the need for long-term tunnelled catheters or implantable devices.
- The care of central venous catheters and portacaths should follow the NHSGGC Vascular Access Procedure and Practice Guideline.
5.4 Selection of Cannulation Site
- When choosing a suitable site for cannulation, both the cannula size and the size and condition of available veins must be taken into consideration. The smallest bore intravenous cannula and the straightest vein, preferably on the back of the hand, should be used.
- Joint flexions should be avoided and in particular the antecubital fossa, especially for vesicants.
- If a vein in the antecubital fossa has to be used this should be flushed with 20mls 0.9% saline prior to bolus administration or infusion. The cannula should be flushed with 30mls of 0.9% saline post administration (weight appropriate volume in infants). The site must be observed throughout the infusion.
5.5 General Guidelines for Handling and Administration of Chemotherapy by Nursing Staff
- Personal Protective Equipment (PPE) –personal protective equipment/PPE should be worn for administration of all SACT. This includes:
- Nitrile Gloves
- Plastic apron
- Armlets
- Goggles/glasses/eye protection
- Face mask when opening capsules, i.e Lomustine
- PPE should be worn when disposing of unused SACT in a purple sharps box, which should be securely closed as per hospital policy and put into the locked waste cupboard in yellow clinical waste bins, which are uplifted twice per day.
- Protective Measures in Pregnancy - Although there is no conclusive evidence that foetal damage occurs when pregnant staff handle cytotoxic agents, individuals should have full access to information and guidance through their line manager and occupational health department.
The decision whether or not to continue working with cytotoxic agents will be at the discretion of the individual and their decision supported by management.
- Spillage - In the event of spillage PPE must be worn, the area sealed off from patients, and the spillage contained (refer to RHC-HAEM-ONC-005). Two pairs of nitrile gloves must be worn, and a spillage kit used to decontaminate the area. This should be disposed of in the clinical waste bin in the locked waste room. Cytotoxic spillage kits are kept in all consulting rooms, the clean utility room in the Schiehallion Daycare Unit and treatment room in the Schiehallion Inpatient Ward. Signage is used to inform staff where spillage kits are located.
In the theatre suite the spillage kit is kept in Theatre 1 utility room and in Outpatients in the treatment room in Clinic 1. The pharmacists must be made aware of the spillage. A DATIX form should be completed and spillage episodes will be audited.
- Extravasation - In the event of extravasation the infusion should be stopped and the extravasation policy followed (RHC-HAEM-ONC-009). Medical and senior nursing staff should be informed. All staff should be aware of where the extravasation kit is kept (in the treatment room in the Schiehallion Ward (inpatients) and the clean utility room in the Schiehallion Daycare Unit - signage in place), and a DATIX should be completed. Extravasation episodes will be audited.
- Disposal - All sharps, containers and bags containing cytotoxic waste must be disposed of in dedicated cytotoxic sharps containers in both the Schiehallion Ward (inpatients) and Schiehallion Daycare Unit (outpatients). All waste is stored in the keypad accessed waste storage room on the second floor. Waste is uplifted twice a day and later incinerated.
- Handling and Disposal of Waste Products i.e. urine, faeces, vomit - Parent/carers handling body fluids are advised by nursing staff to wear nitrile gloves. The disposal of body waste is described in the infection control policy.
5.6 General Comments on Intravenous Administration
- All staff administering SACT must be knowledgeable about the drugs being given, with respect to:
- The appropriate route of administration, according to protocol
- The usual dose ranges for each drug by recipient age
- Possible immediate, short and long term local and systemic side effects
- All SACT doses must be administered by a registered nurse who is trained and assessed as competent in the administration of SACT.
- The administering practitioner must ensure appropriate venous access with regards to:
- Site
- Position
- Patency
- Integrity
- Visibility
- She/he must check the patency of the cannula or central line, by aspirating blood and flushing with 10mls of sodium chloride 0.9% or appropriate solution. SACT should not be administered through a malfunctioning line.
- If there are any doubts regarding cannula patency, the patient must be re cannulated.
- If the placement or patency of a central access device is in doubt, this should be checked by a chest X-Ray and/or linogram prior to commencing treatment.
- The compatibility of the drug with the fluid used for flushing must be checked.
- The patient should be observed for adverse signs and symptoms, and where applicable, managed appropriately.
- The insertion site should be observed during administration of intravenous bolus SACT to detect early signs of drug leakage. For infusional treatment hourly checks must be maintained and recorded.
5.7 Storage and collection of chemotherapy agents
- Intravenous boluses of Vinca Alkaloids must be collected from the aseptic unit by a chemotherapy competent nurse. Other SACT are either collected from the aseptic unit or delivered by a porter. Receipt of all drugs must be recorded on the chemotherapy sheet (see appendix 3) held in the clean utility room.
The practitioner receiving a delivery of chemotherapy must ensure that the SACT is stored in the designated areas at the correct storage temperature.
- In Ward 2B (Schiehallion Daycare) all chemotherapy storage areas will be checked each night and the record sheet signed to confirm that no chemotherapy drugs remain in the department.
- The disposal of unused SACT will be carried out in local areas and disposed of as per policy in the purple lid sharps box, sealed as per waste policy and disposed of in the clinical waste bin in the locked waste room.
- Unused SACT should not be returned to the aseptic unit.
Children with cancer or associated disorders may be referred by primary, secondary or tertiary care. Appropriate investigations to establish the correct diagnosis and stage of disease is the responsibility of the consultant.
Similarly it is the responsibility of the child’s consultant to arrange all necessary investigations to monitor response to treatment, including any toxicities and to investigate suspected or confirmed relapse. Each new patient will be discussed at the disease specific MDT where the diagnosis will be confirmed and treatment agreed. These decisions are recorded on a proforma which is signed off and filed in the patient’s case record. A copy is sent to the patient’s GP. Patients are re-discussed at the MDT at the time of relapse or following any event which might require a change of treatment. They are also discussed at disease specific weekly on-treatment meetings with particular emphasis on toxicities and chemotherapy planning/ordering.
It is the responsibility of each consultant to obtain informed consent for treatment for all patients irrespective of whether the patient is on or off trial.
As per CEL 30 (2012) all staff involved in SACT must have the appropriate skills, knowledge and training in their field of practice. Evidence of this training is documented in each staff member’s training record and must include the following:
- principles of safe use and relevant national guidance
- local policy and procedures on safe use
- principles of SACT
- CMGs and SACT protocols relevant to area of clinical practice
- consent and information giving
- holistic assessment of patients receiving SACT
- prevention and management of adverse effects
- selection and use of equipment
- safe handling of cytotoxic chemotherapy
It is also a consultant responsibility to allocate treatment and to ensure that the relevant up to date treatment protocol is available. Copies of all protocols and guidelines are maintained by data management.
All children are treated on NCRI trials if these are open for their particular disease. In the absence of a clinical trial they will be treated on a national guideline. The exceptional child with an extremely rare disorder may be treated according to best practice. The diagnosis and treatment with expected benefits, outcomes and risks are explained to the parents/child.
Written trial specific and age-appropriate information is given to the parents/patient and following an appropriate period of time to allow parental/patient understanding, written consent is obtained. Consent will be documented on a generic consent form for all patients and in addition on a trial specific consent form if recruited to a clinical trial. The site file for each trial will list the medical practitioners who can take consent. Patients/parents/carers for whom English is not their first language will be offered the services of an interpreter at the time of diagnosis, when consent is taken, and later as required.
All patients will be assessed by a doctor or ANP immediately prior to receiving chemotherapy. This assessment will include general fitness for chemotherapy, performance status (PS), necessary critical tests and the presence of associated toxicities.
The results of any clinical test required prior to chemotherapy commencing, PS and grading of any toxicity / co-morbidity MUST be completed and signed off by the doctor/ANP prior to authorising any new course of chemotherapy. Haematological toxicity does not have to be completed for patients with acute leukaemia/lymphoma during maintenance treatment when blood counts are reviewed regularly and doses of chemotherapy titrated accordingly
In the very rare circumstance where a chemotherapy trained doctor or ANP is not available to authorise a patient’s treatment a member of the Schiehallion medical team can sign the authorisation, but only after discussion with a Consultant. This must be clearly documented on the prescription and the entry must be counter-signed by the Consultant at the earliest opportunity.
The medical practitioner will have received structured education on the principles of chemotherapy. They will have completed induction training within the unit and will have been deemed competent prior to prescribing cytotoxic drugs. Only consultants, speciality doctors and trainees of ST4 level and above can prescribe chemotherapy and only after appropriate training.
This SOP assumes that local practices and policies for education are implemented and carried out within the haematology/oncology unit and that adherence to these will be regularly monitored, audited and updated.
The individual practitioner must be familiar with, and adhere to, related policies and protocols noted in section 8 – Related Documentation.
Intrathecal cytotoxic drugs will only be administered by doctors of ST4 level and above, staff in non-Consultant Career Grades and Consultants who have Division certification and are listed on the intrathecal register.
NB: Doctors in training will not administer bolus vinca alkaloids by peripheral venous access. This will only be done by SACT Intravenous bolus trained nursing staff.
Written informed consent for treatment must be taken at an appropriate period of time after the patient/parent has been provided with verbal and written information, which includes the potential risks and anticipated benefits. This information should be provided by the Paediatric Consultant Haematologist or Oncologist responsible for starting or initiating the next course of SACT and, following this discussion, the Consultant should sign off Page 3 of the generic consent form (HAEM-ONC-TEMP-002).
6.1 Initial Treatment Decision and Consent
- The initial decision to treat a patient with SACT and the selection of the SACT protocol must be made by a Paediatric Consultant Haematologist or Oncologist.
- Informed and written consent must be obtained after relevant verbal and written information have been provided. If, following the appropriate period of time, consent is taken by a delegated depute then they must sign off Page 2 of the generic consent form (HAEM-ONC-TEMP-002) post Page 3 being signed off by a Consultant.
- Delegated deputies will only be:
- Specialty doctors
- Associate specialist
- Adult haematology trainees
- GRID oncology trainees
- The decision to treat must be documented in the patient notes.
- The treatment plan, a copy of the treatment protocol and flowsheets (signed and dated by Consultants) must also be filed in the patient’s extended scanning record prior to SACT administration. This information must also be communicated to the GP within 14 days.
- The performance status and any co-morbidities at diagnosis must be documented in the patient’s notes. In patients with a poor performance status the rationale for treatment should be clearly documented as well as any additional monitoring arrangements in a patient specific treatment plan.
- The protocol or guideline must be clearly documented in the patient’s records by the Paediatric Haematologist or Oncologist.
- A copy of consent to chemotherapy must be scanned onto Clinical Portal.
- Where electronic case records are being used a summary of the diagnosis, treatment protocol and associated supportive care must be added to the appropriate e-form on Clinical Portal.
6.2 SACT Protocol
- The treatment protocol or guideline should contain the following information:
- Eligibility and exclusion criteria
- Drugs, doses, scheduling, number of cycles and supportive care
- Dose modifications
- Critical tests required prior to each course or cycle
- Potential toxicities
- Frequency of disease evaluation
6.3 Prescribing
Only a doctor of ST4 and above who has completed the appropriate medical training on chemotherapy can prescribe SACT, with the exception of Hydroxycarbamide which can be prescribed by named nurse prescribers:
- Lyn Docherty and Ruth Bissell, Haematology Nurse Specialists
- Wendy Taylor, Advanced Nurse Practitioner
- SACT prescribers must have access to all relevant protocols and guidelines to support safe and appropriate prescribing
- SACT prescribers must have access to relevant individual patient information on previous modifications to SACT prescriptions
- The patient must be assessed for adverse effects at appropriate intervals as determined by the trial protocol. These must be graded using CTCAE criteria and documented appropriately on either the chemotherapy template prescription or on the toxicity tab on Chemocare. In addition required adverse events, SAEs and SARs should be reported to the sponsor as directed in the protocol.
All unexpected adverse drug reactions should be reported to www.mhra.gov.uk/yellowcard . In addition, all adverse events occurring in patients not treated on trial will be discussed at the daily handover and the event and management of the SAE will be recorded in the patient’s casenotes. Those requiring treatment modification will be recorded on the treatment plan within the shadow notes.
- The performance status must be assessed and recorded prior to each course of chemotherapy
- Oral maintenance therapy can only be prescribed by appropriately trained medical staff at ST4 level and above, staff in a non consultant career grade and Consultants
- Intravenous courses of infusional and bolus chemotherapy can only be prescribed by medical staff at ST4 level and above, staff in non-Consultant Career Grades and Consultants
- Intrathecal chemotherapy can only be prescribed by ST4 level and above, staff in non-Consultant Career Grades and Consultants currently on the intrathecal register
- SACT is prescribed using Chemocare or a standardised, validated template prescription wherever possible. All SACT prescriptions should comply with current legal requirements and local prescribing policies. Prescriptions must be written in a clear and unambiguous manner and include:
- name, date of birth, CHI
- recent weight (two weights and heights if first course)
- diagnosis
- name of protocol
- cycle number and date of intended treatment
- performance status and toxicity score at the start of each course
- relevant critical tests
- generic names of each drug and calculated doses to be administered
- route and duration of administration
- diluents and infusion volumes where appropriate
- hydration schedules and pre-medication if required
- appropriate supportive care
- a record of any dose modifications
- indication of concomitant radiotherapy where applicable
- signature of prescriber and date prescription is written or generated (these may be electronic if on Chemocare)
- signature documenting pharmaceutical verification and date
- signature documenting administration and date
NB: The initial decision to administer cytotoxic chemotherapy is made by an accredited haematology/oncology consultant or an appropriately trained and competent nominated deputy and the decision must be CLEARLY recorded in the patient’s CLINICAL notes.
NB: A validated electronic chemotherapy prescribing system is operating and under ongoing DEVELOPMENT WITHIN the Schiehallion Unit. All protocols will in time be available electronically. IN THE EVENT OF A SYSTEM FAILURE, STAFF WILL REVERT TO PAPER TEMPLATE PRESCRIBING
6.4 Out of hours
In the absence of medical staff of appropriate grade on site, minor changes to prescribed/scheduled intravenous chemotherapy (such as date/time or pharmacy instituted rounding up or down of doses) can be made by ST3 medical staff after authorisation from the consultant on call and in consultation with the nurse in charge of the unit. Treatment must be countersigned on the ward chart the next day by the consultant.
In the exceptional circumstances that chemotherapy is required out of hours, the requesting consultant will contact the on-call pharmacist who will liaise with a Cancer Care Pharmacist and the out of hours Pharmacy Aseptic Team as per the GG&C Out of Hours Supply of SACT policy.
6.5 Deferring of Chemotherapy
It is very important to record the reason for any deferral of chemotherapy courses. This should be documented in the appropriate section at the front of the chemotherapy template prescription or electronically on ChemoCare. The reason for the deferral and the next intended treatment date must be recorded.
If any chemotherapy course is delayed for more than 7 days the patient’s consultant must be informed. If a deferral results in a modification to the intended course of treatment the prescription must be amended appropriately or rewritten. Any change to a prescription must be clearly annotated and signed by an appropriate chemotherapy prescriber and the reason documented on the prescription. Any major change must also be documented on the patient treatment plan. Any modified/rewritten prescriptions must be given to the appropriate clinical pharmacy team for rescreening.
Any deferral of chemotherapy must be completed on the appropriate prescription (template or Chemocare) by a member of the clinical team who has reviewed the patient and made the decision to defer. Within the Schiehallion Ward this must be a chemotherapy trained member of medical staff or ANP. Within DCU this can be a chemotherapy trained member of medical staff, ANP, senior nurse in charge or paediatric oncology outreach nurse specialist.
Pharmaceutical Verification, Preparation and Dispensing of SACT
- Receipt of prescriptions:
- Intrathecal
Refer to GG&C IT policy and RHC-HAEMONC-013 - Oral & Parenteral
- All prescriptions for chemotherapy requiring aseptic dispensing must be presented to a paediatric haematology/oncology pharmacist or confirmed on Chemocare by 12 noon at the latest on the day chemotherapy is required and by 12noon on a Friday for weekend chemotherapy. The final clinical authorisation for these prescriptions must be communicated to a paediatric haematology/oncology pharmacist at the latest by 3pm for preparation the same day and 3pm on a Friday for the weekend.
- All confirmed and authorised prescriptions for oral dispensing must be presented to a paediatric haematology/oncology pharmacist by 4pm at the latest for dispensing the same day. No prescriptions for oral SACT will be dispensed at the weekend unless previously verified by a paediatric haematology/oncology pharmacist.
- Intrathecal
- Verification of SACT prescriptions
- All prescriptions for SACT and the associated supportive care must be verified by pharmacists experienced in paediatric haematology and oncology before any aseptic or oral chemotherapy dispensing can take place.
This may be a two step process where the initial confirmed (ie the intended treatment) prescription is presented to pharmacy in advance and is screened and forwarded for pre-planning aseptic or oral chemotherapy dispensing. The patient may be reviewed later by suitably trained medical staff and/or advanced nurse practitioners who give final authorisation to a paediatric haematology/oncology pharmacist before any SACT is issued for that patient. The signature of the individual pharmacists involved in each stage of the verification must be clearly documented and dated on the original prescription. - Prior to any SACT being fully authorised the paediatric haematology/oncology pharmacist must verify the prescription which includes carrying out the key pharmaceutical checks detailed below:
- The medical prescriber’s details and signature are present and they are suitably trained and authorised to prescribe SACT
- The approved treatment protocol or guideline for that patient is documented and is available for review
- An appropriate consultant has documented the patient specific treatment plan for the first cycle or course of SACT and this is appropriate for both the indication and patient SACT’s history
- There are no known drug allergies, previous adverse events or food interactions which would affect the intended treatment
- The treatment is scheduled appropriately in relation to the interval since previous therapy
- The patient demographics are correct and include as a minimum the patient name, date of birth and CHI number
- The patient weight which has been used to calculate the doses must be recorded and checked. For the first cycle, two independent weights and heights must be available. These will be used to calculate BMI and the results are plotted on an appropriate growth chart to ensure that the patient is between the 2nd and 98th percentile. For children and teenagers with a BMI that falls within the 2nd and 98th percentile, the dose will be calculated using actual weight by SAPE body surface area calculation or charts. For children and teenagers who have a BMI >98th percentile or <2nd percentile the dosing weight must be discussed and agreed by the patient’s consultant and reviewed at regular intervals. Patients must be weighed at each course of treatment and this weight will be used to calculate the doses for the subsequent cycle. The patient will be reweighed prior to final authorisation, however doses will only be changed if the change in weight results in a >10% change in actual dosing or if a dose modification is indicated
- All dose calculations and units are correct and have been calculated according to the treatment protocol or guideline
- Cumulative doses and maximum individual doses are appropriate
- The reason for any dose modification is documented and the dose adjustment is appropriate
- The method of administration is appropriate
- The relevant laboratory values are within acceptable ranges as defined in the treatment protocol or guideline
- Other essential tests eg audiology, ECG etc have been undertaken and are satisfactory
- Drug doses are appropriate with respect to renal function, hepatic function and other co-morbidities
- Supportive care is prescribed and is appropriate for the patient and the SACT
- Dose modifications / appropriate antimicrobial prophylaxis and any other prophylactic measures
- to minimise risk of adverse events
- The paediatric haem/onc pharmacists must clarify with both the aseptic unit and oral dispensing service any complex or unusual SACT prescriptions
- The paediatric haematology/oncology pharmacists are responsible for liaising with both the aseptic and oral dispensing service in a timely manner with regards to final verification or deferment of SACT prescriptions. The initial plan for treatment is communicated via an electronic daily list, however, final verification and deferment is by telephone and the electronic list updated
- Any dose modifications which are made to an existing SACT prescription must be verified using the same process as the initial prescription
- If the prescription is for a therapy which is not on an approved clinical guideline or clinical trial protocol the case must be discussed with the responsible consultant. Any publications or e mail correspondence to support the choice of treatment must be available and the paed haem/onc pharmacist must be satisfied that the prescription is appropriate for the individual patient
- The requirement for dose modifications and/or appropriate prophylaxis to minimise the risk of neutropenic sepsis and other predictable adverse events
- All prescriptions for SACT and the associated supportive care must be verified by pharmacists experienced in paediatric haematology and oncology before any aseptic or oral chemotherapy dispensing can take place.
- Pharmacy Documentation
- A pharmacy chemotherapy treatment schedule is maintained for all patients receiving SACT. These contain a copy of the patient demographics, relevant growth charts, treatment details extracted from the treatment protocol or guideline, a summary sheet of dates of SACT administration and any relevant additional information. These records are held in the Schiehallion Ward Pharmacists’ Room.
- All inpatients receiving SACT are reviewed daily Monday to Friday by a paediatric haematology/oncology pharmacist and a pharmaceutical care plan is available on Clinical Portal. Out-patients receiving SACT are reviewed prior to SACT administration and relevant pharmaceutical information is documented on the pharmacy chemotherapy treatment schedule.
Additional information on supportive care is documented on the pharmacy generic continuation sheet on Clinical Portal.
- Aseptic Unit Responsibility
- The Lead Pharmacist for Preparative Services for GG&C is responsible for ensuring the aseptic unit comply with the following:
- SACT will only be supplied against a verified prescription
- All SACT requiring aseptic manipulation is prepared in accordance with local standard operating procedures, legislative requirements, national standards and guidelines
- Systems are in place to independently audit the aseptic service every two years and non-conformance remedial plans are in place with assigned timescales
- All SACT is dispensed and labelled for the individual patient in a ready to administer form
- Changes being made to drug doses will not be accepted unless these are verified by a paed haematology/oncology pharmacist
- Training of any non-clinical staff involved in the transport of SACT
- Intrathecal SACT is only issued directly to medical staff on the paediatric register who are performing the procedure and this issue is documented
- Vinca alkaloids are only issued directly to nursing staff and this is documented
- Dispensing of Oral SACT:
- SACT will only be supplied against a verified prescription
- All oral SACT is dispensed in accordance with local standard operating procedures, legislative requirements, national standards and guidelines
- All oral SACT is dispensed and labelled for the individual patient for a specified course of treatment
- Only changes to drug doses which have been verified by a paediatric haematology/oncology pharmacist will be accepted
- Storage of SACT on within the Schiehallion Ward and Schiehallion Daycare Unit:
- Only SACT dispensed for an individual patient will be kept and stored at ward level
- Oral SACT prescriptions for in-patients are stored in the patient’s own locked POD lockers and are issued from there by pharmacy and/or nursing staff on discharge. These POD lockers are only accessible by pharmacy and nursing staff. Prescriptions for out-patients are stored in a locked dedicated chemotherapy cupboard and/or fridge and are issued by pharmacy or nursing staff directly to the patient or carer
- The receipt of parenteral chemotherapy issued by the Aseptic Unit must be documented on the appropriate Record of Chemotherapy Issued sheet (see appendix 3). Parenteral SACT must be stored in the appropriate dedicated chemotherapy cupboard or refrigerator on both the Schiehallion Ward and Schiehallion Daycare Unit until it is required for administration to the patient. The nurse administering the SACT must sign this out from the appropriate record of issue. SACT is issued by the Aseptic Unit daily and, therefore, only doses required in the next twenty four hour period will be available in the ward area
- Any unused SACT treatment must be disposed of appropriately in a specific cytotoxic sharpsafe container. The disposal must be documented on the record of issues sheet
- The Lead Pharmacist for Preparative Services for GG&C is responsible for ensuring the aseptic unit comply with the following:
- Training of Clinical Pharmacist working in Paediatric Haematology/Oncology
- The clinical pharmacists receive fully supervised training and only when deemed competent by a senior paediatric haematology/oncology pharmacist will they be allowed to perform any individual role unsupervised. Within the unit they are also encouraged to attend all multidisciplinary education sessions and on-treatment meetings.
- This SOP assumes that general training within paediatric practice has already been delivered as part of their hospital induction training.
- Patient Counselling and Patient Information Leaflets
- All patients receiving oral SACT at home must be counselled by a paediatric haematology/oncology pharmacist prior to any treatment commencing. A drug specific patient information leaflet is issued at this time. The counselling covers safe storage of SACT in the home, safe administration, dosing information, how to obtain supplies of oral syringes, if required, and any common side-effects. Patients/parents are also given the contact details of a paediatric haematology/oncology pharmacist should they require any additional pharmaceutical information. With each additional SACT dispensing counselling will be repeated if necessary. Patients are counselled to return all unused supplies of SACT at their next hospital visit to monitor compliance and ensure appropriate disposal, with the exception of the leukaemia clinic where families are advised to return home supplies on Weeks 1, 5 and 9 of each treatment cycle.
- Availability of SACT Outwith Normal Working Hours
- In the rare and exceptional clinical case where SACT is required out of hours or over a weekend or public holiday, the consultant in charge must make this request via the on-call paediatric pharmacist.
- Refer to the GG&C Policy for the Out of Hours Supply of SACT for further detail.
- The reasons for requesting chemotherapy out of hours must be clearly documented in the patient’s case records by the Consultant.
- The SACT prescription for the out of hours chemotherapy must be signed by the Consultant and verified by a paediatric haematology/oncology pharmacist prior to commencing and dispensing.
NB: If Chemocare fails the contingency plan is that standard prescription templates on Q pulse will be used. If the internet access fails then electronic back-up templates on the pharmacy server will be used. If all IT fails a paper-based generic prescription sheet will be used.
This SOP will be reviewed every two years.
This procedure is subject to external audit under the Intrathecal Cytotoxic Chemotherapy Administration checklist contained within the CEL 2009 (21) guidance. Audits are normally performed every 3 years.
There are no exceptions to this SOP and deviations are not acceptable.
Appendix 1: reporting structure
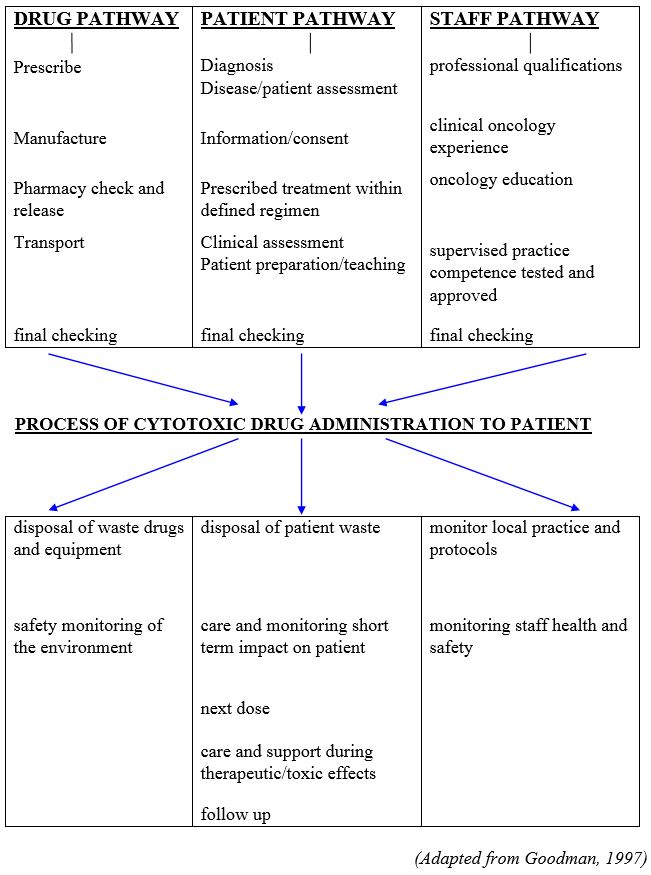
- KC Hine D (1995). The Calman Hine Report: A Policy Framework for Commissioning Cancer Services. Department of Health, London
- Carmignani SS Raymond GG (1997). Safe handling of cytotoxic drugs. Oncology Nursing Forum; 24 (1 suppl) 41-48
- DOH (1997). Commissioning Cancer Services in Scotland: Guidance on Pharmaceutical Services and Nursing Services. Scottish Office Department of Health, Edinburgh
- Eli Lilly & Co. Cytotoxic Chemotherapy: Guidelines for the administration of Cytotoxic Drugs and The Nursing Care Of Patients receiving Chemotherapy, 5th Ed. Eli Lilly, Basingstoke
- Goodman I (1997). National clinical guidelines for the administration of cytotoxic chemotherapy (Draft copy)
- Grundy M (1998). Chemotherapy Administration by Nurses: An Audit of Practice and Educational Preparation. Report and Executive Summary, National Board for Nursing, Midwifery and Health Visiting for Scotland, Edinburgh
- Health and Safety Executive (1983). Preparation for the Safe Handling of Cytotoxic Drugs. Guidance Notes, MS;21, HMSO, London
- Health and Safety Executive (1995). The Control of Substances Hazardous to Health Regulations. Statutory instrument No. 1657, HMSO, London
- Nieweg RMB de Boer, M Dubbleman RC, Gall HE, Hesselman GM, Lenssen PCHP, van Maanen LWGM, Majoor PMFM, Ouwerkerk J, Slegt JH (1994). Safe handling of antineoplastic drugs - Results of a survey. Cancer Nursing; 17, 6, 501-511
- Royal College of Cancer Nursing Society (1989). Safe Practice with Cytotoxics, Scutari, Middlesex
- Scottish Executive Health Department. Guidance for the safe administration of cytotoxic chemotherapy. HDL (2005) 29
- Scottish Executive Health Department. Safe administration of intrathecal cytotoxic chemotherapy. HDL (2004) 30
Last reviewed: 01 April 2022
Next review: 30 April 2024
Author(s): Prof Brenda Gibson
Version: 12
Approved By: Schiehallion Clinical Governance Group
Document Id: RHC-HAEM-ONC-014

