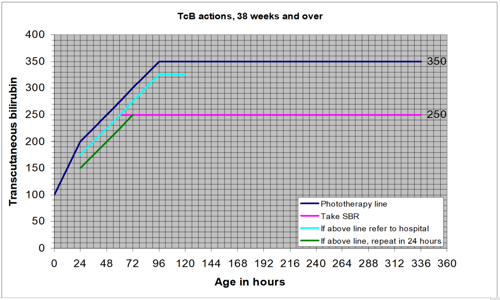
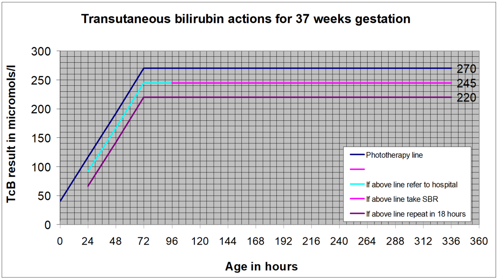
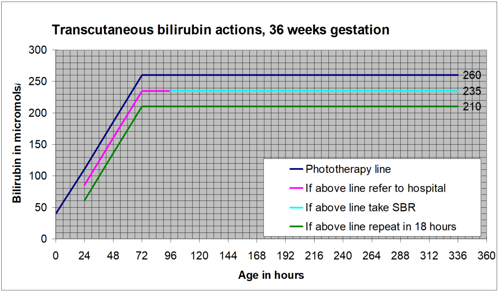
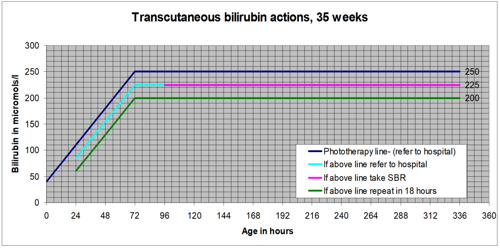
Jaundice can develop at any time over the first few days of life. All possible opportunities should be used to look for jaundice over this time.
When looking for jaundice (visual inspection):
- Check the naked baby in bright and preferably natural light
- Examine the sclerae and gums, and press lightly on the skin to check for signs of jaundice in ‘blanched’ skin
- Be aware that hyperbilirubinemia may be harder to see visually in darker skin
Assess for jaundice at every interaction with a newborn baby in the first days of life. Ensure adequate lighting. Document the absence or presence of jaundice whenever writing a clinical note. In the “colour” section of the daily check in Badgernet the presence or absence of jaundice should be noted each day.
Particular attention must be paid to the following groups:
- gestational age under 38 weeks
- a previous sibling with neonatal jaundice requiring phototherapy
- mother's intention to breastfeed exclusively
- visible jaundice in the first 24 hours of life.
Presence of these risk factors should be noted on admission to the ward and it should be ensured that they are regularly assessed for jaundice. NICE recommend that babies with the above factors associated with an increased likelihood of developing significant hyperbilirubinaemia receive an additional visual inspection by a healthcare professional during the first 48 hours of life.