Hypertension in children and young people, Renal Unit RHC
exp date isn't null, but text field is
Objectives
The following guideline has been developed and is regularly reviewed by clinicians within the Renal Unit at the Royal Hospital for Children, Glasgow. It is based on current evidence and best practice relating to the investigation and management of hypertension in infants, children, and adolescents (aged 1-17 years). A separate West of Scotland guideline exists for ‘Hypertension in neonatal patients’ and this should be referred to if necessary.
This guideline is intended for use by clinicians, nursing staff, and pharmacists. For further discussion of this guideline, please contact a member of the nephrology team based within the Renal Unit (On-call Renal Consultant through switchboard, Renal Registrar on page 18282 or 84563, or via the Renal Hot Desk on 0141 452 4563).
Blood pressure (BP) rises throughout childhood relative to age and height. As with height and weight, there are specific percentiles for BP measurement available for both genders. Published BP values for males and females are included in Appendices 1 and 2 respectively.
The 2016 European Society of Hypertension guidelines for the management of elevated BP in children and young people (CYP) classifies hypertension according to centile values up until 16 years of age (Table 1) (1).
Primary (or essential) hypertension, where no identifiable cause is found, is a diagnosis of exclusion in children. Children <6 years of age with elevated BP are more likely to have an established cause (secondary hypertension).
Table 1 – Classification of hypertension below and above 16 years of age

Severe hypertension may be referred to as a ‘hypertensive crisis’. This is a term that traditionally includes both ‘hypertensive urgency’ and ‘hypertensive emergency’. Hypertensive urgency is defined as severe BP elevation without progressive target-organ damage whereas hypertensive emergency is defined as severe BP elevation with impending or progressive target-organ damage. Realistically, any extreme elevation of BP where there are concerns about the child’s symptoms should prompt the initiation of treatment and very close monitoring of BP with the aim of avoiding any precipitous fall. Dropping BP too quickly raises the risk of inducing autoregulatory responses in key circulatory beds in the brain, heart, and kidneys. This may lead to irreversible target-organ damage, including permanent neurologic sequelae, visual defects, myocardial infarction, and renal impairment.
BP is now measured using automated oscillometric devices (e.g., Dinamap® device) rather than mercury sphygmomanometry. While convenient and widely used, if a raised BP is suspected following use of an automated device, auscultation using a validated manual device (e.g., Accoson Greenlight® sphygmomanometer) must be performed to confirm a suspected raised BP. The British and Irish Hypertension Society keep an updated list of devices validated for use which is available at: https://bihsoc.org/bp-monitors/for-specialist-use.
An appropriate-sized cuff must be used. The width of the cuff should cover at least 75% of the upper arm from the acromion to the olecranon, leaving sufficient space at the antecubital fossa to allow application of the bell of the stethoscope. The widest cuff possible that covers the upper arm should be used. The cuff bladder length must encircle and cover 80-100% of the arm circumference.
Measure the BP with the child in a seated position and their arm gently supported, ideally after the child has been sitting quietly for 3 minutes (or lying supine for an infant). BP cannot be reliably assessed if the child is upset/crying. Ideally, hypertension should be confirmed with an average of three measurements on three separate occasions/days/visits. Measure height and plot on an appropriate gender-specific growth chart to establish the height percentile then refer to the BP centile chart for the appropriate gender. The normative values for BP in children are based on right arm measurements, and BP values measured from the leg should be interpreted with extreme caution. If an isolated right arm BP is found to be elevated, then BP should also be assessed in all 4 limbs to help exclude evidence of coarctation. In coarctation, the BP in the lower extremities is expected to be lower, or sometimes unmeasurable, compared with that of the right arm. The left arm BP may also be elevated if the origin of the left subclavian artery is proximal to the coarctation (occurring in most cases).
Korotkoff phases (for manual assessment of BP):
- K1 corresponds to the systolic BP
- K2 is initial disappearance of the tapping sound
- K3 is return of the tapping sound
- K4 is muffling of the tapping sound, and this corresponds with the diastolic BP in those up to 13 years of age
- K5 is complete disappearance of the tapping sound, and this corresponds with diastolic BP after 13 years of age
- In some children, Korotkoff sounds can be heard down to 0 mmHg, and this excludes diastolic hypertension
Doppler ultrasound assessment of BP is recommended for younger children (particularly <5 years) and for those who are overnight or obese. A Doppler probe is held over the radial or brachial pulse and the BP cuff should be inflated to 20-30 mmHg above where the pulse is no longer audible. The cuff should then be deflated at a rate of 2-3 mmHg per second and the systolic pressure is evident when there is a Doppler phase shift. Doppler assessment cannot be used for the measurement of diastolic BP.
24-hour ambulatory blood pressure monitoring (ABPM) is increasingly recognised as useful in the diagnosis and management of hypertension in CYP. It can only reliably be performed in older children (>5 years old) with a minimum height of 120 cm. Normative values for ABPM thresholds exist (2, 3) and the Renal Unit at RHC have an ABPM request form (Appendix 3) and proforma for reviewing and interpreting ABPM data (Appendix 4). At RHC, the request form should be submitted to Ward 1C and the ABPM monitor is fitted there. Families are then expected to return the following day for removal of the monitor. The report is then uploaded and sent back to the referrer for interpretation. Infants and younger children may require admission to hospital for BP monitoring to confirm the diagnosis as reliable BP recordings can be difficult to obtain in this age group. In other units, speak to your SPIN nephrology lead to see if ABPM can be performed locally.
Indications for the use of ABPM in the diagnosis of hypertension include (4):
- Confirming hypertension before drug treatment or assessing BP whilst on antihypertensive treatment
- Diagnosing white coat hypertension (reported in up to 50% of cases suspected on casual BP readings) (5)
- Diagnosing nocturnal hypertension
- Chronic kidney disease (including patients on dialysis)
- Types 1 and 2 diabetes mellitus
- Renal, liver or heart transplant recipients
- Severe obesity +/- obstructive sleep apnoea (OSA)
It may be important to ask about the following:
- Signs/symptoms associated with elevated BP:
- Nausea ± vomiting
- Headaches
- Visual disturbance
- Behavioural change
- Altered mental state
- Drowsiness
- Seizures and focal neurology
- Epistaxis
- Abdominal pain
- Cardiac symptoms (e.g., chest pain and palpitations)
- Episodes of flushing (along with palpitations this could be a sign of phaeochromocytoma)
- Oedema (periorbital and peripheral) and dyspnoea secondary to congestive heart failure
- Weight faltering or weight loss
- Intermittent claudication in lower limbs and poor exercise tolerance (may be a sign of mid-aortic syndrome with vascular insufficiency to lower extremities)
- Snoring (as a manifestation of OSA)
- Symptoms suggestive of an underlying renal disorder including polyuria, oliguria/anuria, nocturnal enuresis, and haematuria
- Neonatal history:
- Prematurity
- Umbilical catheterisation (a risk for renal artery and renal vein thrombosis)
- Bronchopulmonary dysplasia / chronic lung disease
- Persistent patent ductus arteriosus
- Past medical history of kidney disease and urinary tract infections:
- Renal parenchymal disease and scarring is a major risk for hypertension
- Congenital heart disease (e.g., coarctation) and cardiovascular risk factors
- Family history:
- Primary hypertension
- Systemic disease, particularly vasculitis
- Endocrine disorders (e.g., Cushing syndrome and hyperthyroidism)
- Sudden death, kidney failure, myocardial infarction, ± cerebrovascular event
- Drug history:
- Steroids
- Other immunosuppressant agents e.g., tacrolimus/ciclosporin
- Decongestants e.g., phenylephrine
- Stimulant medications e.g., methylphenidate
- Oral contraceptives
- Illicit drugs e.g., ecstasy and amphetamines
- Tobacco
- Alcohol
- Vitamin D (intoxication)
- Recent discontinuation of antihypertensive agents
- Detailed dietary history (including salt intake) and physical activity history. Recommended salt intake for 4-6 years of age is 3 g/day; for 7-10 years of age is 5 g/day; and for ≥11 years is 6 g/day
- Signs of heart failure:
- Tachycardia
- Gallop rhythm
- Hepatomegaly
- Raised jugular venous pressure
- Absent of weak femoral pulses, or brachial/radial-femoral delay, potentially indicative of coartaction of the aorta. If detected, then measure difference between four limb BP
- Neurological features:
- Weakness
- Hypotonia or hypertonia
- Hyper-reflexia and clonus
- Upgoing plantars
- Cranial nerve deficits
- Papilloedema and/or retinal haemorrhages
- Organomegaly and/or abdominal masses
- Evidence of thyroid disease (e.g., goitre and eye signs)
- Carotid, abdominal and/or femoral bruits suggestive of arterial narrowing
- Obesity and Cushingoid features
- Ambiguous genitalia as seen in androgenital syndromes
- Neurocutaneous features such as café-au-lait spots and axillary freckling (as seen in neurofibromatosis type 1), or ash leaf spots and adenoma sebaceum (as seen in tuberous sclerosis)
- Signs suggestive of underlying chronic kidney disease:
- Short stature
- Bony disease (e.g., rickets)
- Evidence of anaemia
Table 2 highlights various potential causes of hypertension, subdivided into primary hypertension and secondary hypertension. Please note that this list is not exhaustive.
Table 2 – Potential causes of hypertension
|
Primary (essential) hypertension |
|---|
|
Obesity |
|
High salt diet |
|
Family history of hypertension and/or cardiovascular disease |
|
Secondary hypertension in neonates and infants |
|
Renal causes:
|
|
Cardiac causes:
|
|
Bronchopulmonary dysplasia / chronic lung disease |
|
Raised intracranial pressure |
|
Obstructive sleep apnoea (OSA) |
|
Previous extracorporeal membrane oxygenation (ECMO) |
|
Pain |
|
Secondary hypertension in children and adolescents |
|
Renal causes:
|
|
Cardiac causes:
|
|
Endocrine causes:
|
|
Neurological causes:
|
|
Monogenic (single gene) disorders – may present in infancy:
|
|
Drug-related causes (as listed in ‘History’ section above) |
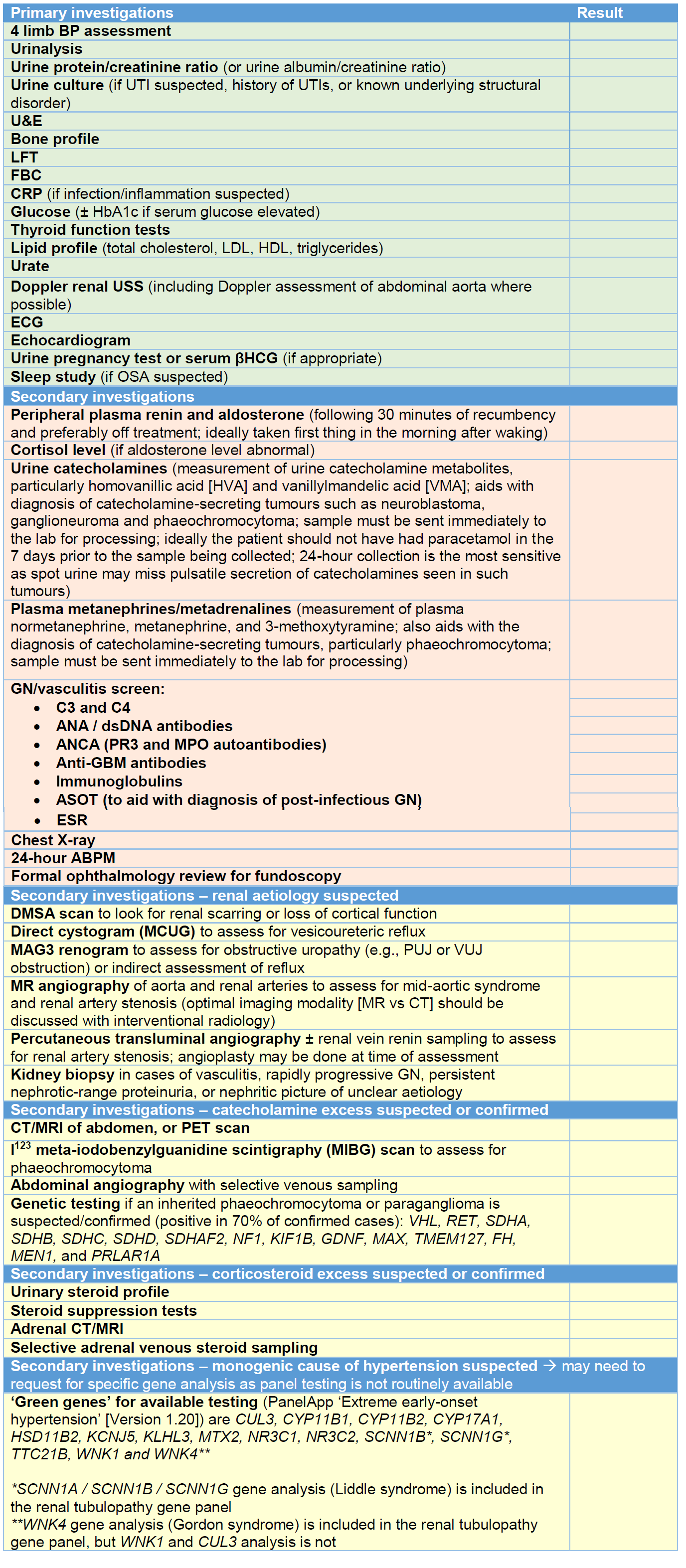
The extent to which hypertension is investigated depends on its severity and the information obtained from a careful history and examination. A family history of hypertension, kidney disease or endocrine causes may help guide the clinician in a particular direction. Table 3 highlights primary and secondary investigations. Primary investigations are essential in any CYP with suspicion of secondary hypertension and, where possible, these investigations should have been conducted/arranged before discussion with nephrology. Secondary investigations may be arranged after discussion with nephrology, or by the nephrology team themselves.
Table 3 – primary and secondary investigations.
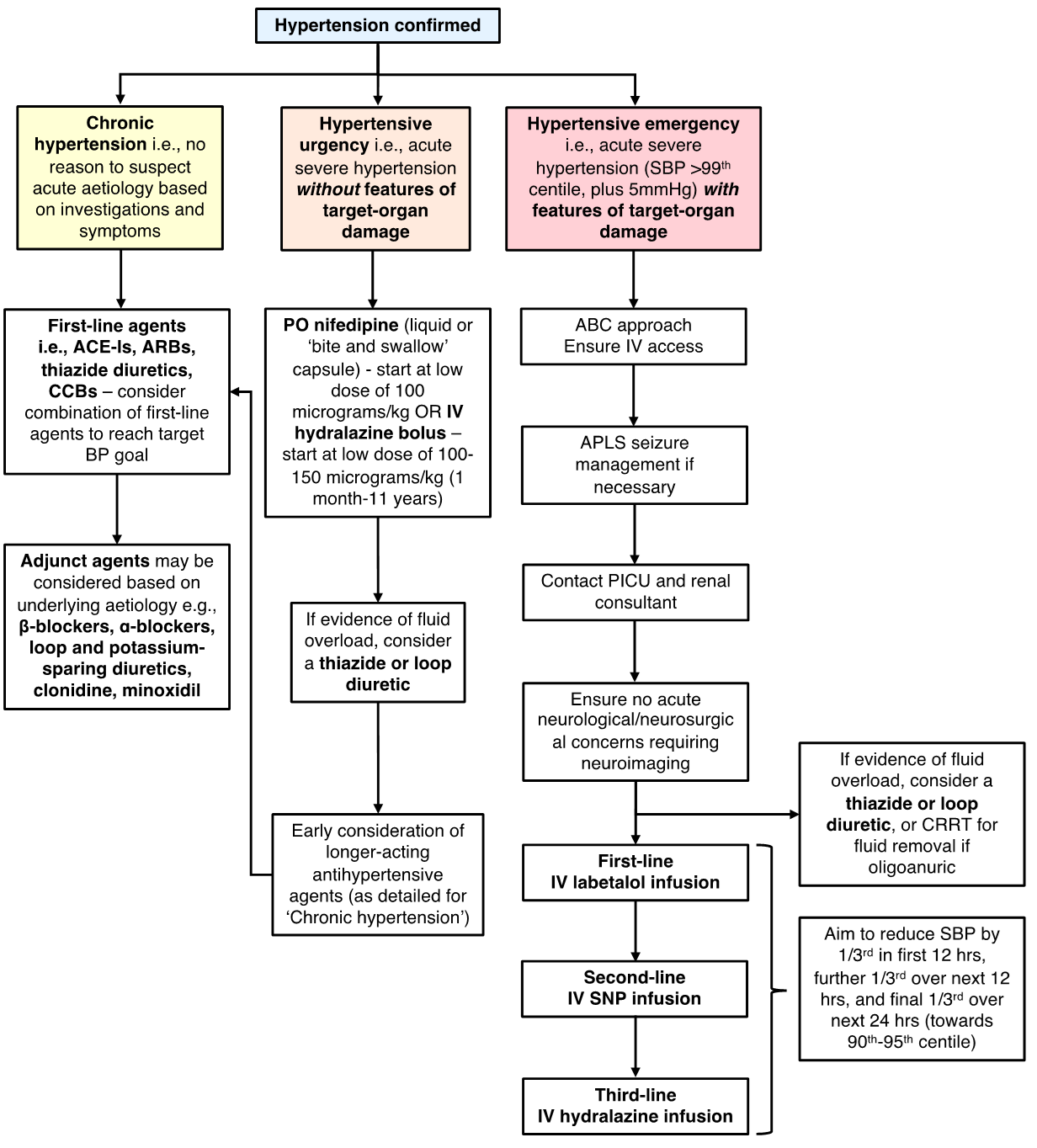
Choice of treatment will depend on whether hypertension is primary or secondary in nature. It will also depend on the severity of the hypertension and whether there are more concerning features (i.e., hypertensive emergency or crisis).
Management of primary hypertension
Treatment of primary hypertension involves lifestyle modification. The main aim is to increase energy expenditure, reduce energy intake, and limit sodium intake. Increased intake of fresh fruit and vegetables, along with low-fat dairy products, has been demonstrated to have a positive impact on BP control in CYP (6). Other beneficial measures include avoidance of caffeine (including energy drinks), alcohol and smoking. Realistically, pharmacological treatment may be initiated to help control BP whilst lifestyle modifications are implemented as it can take time before any discardable benefit becomes apparent.
Management of secondary hypertension
- Management of chronic hypertension:
- Choice of medication will be guided by the underlying disease process.
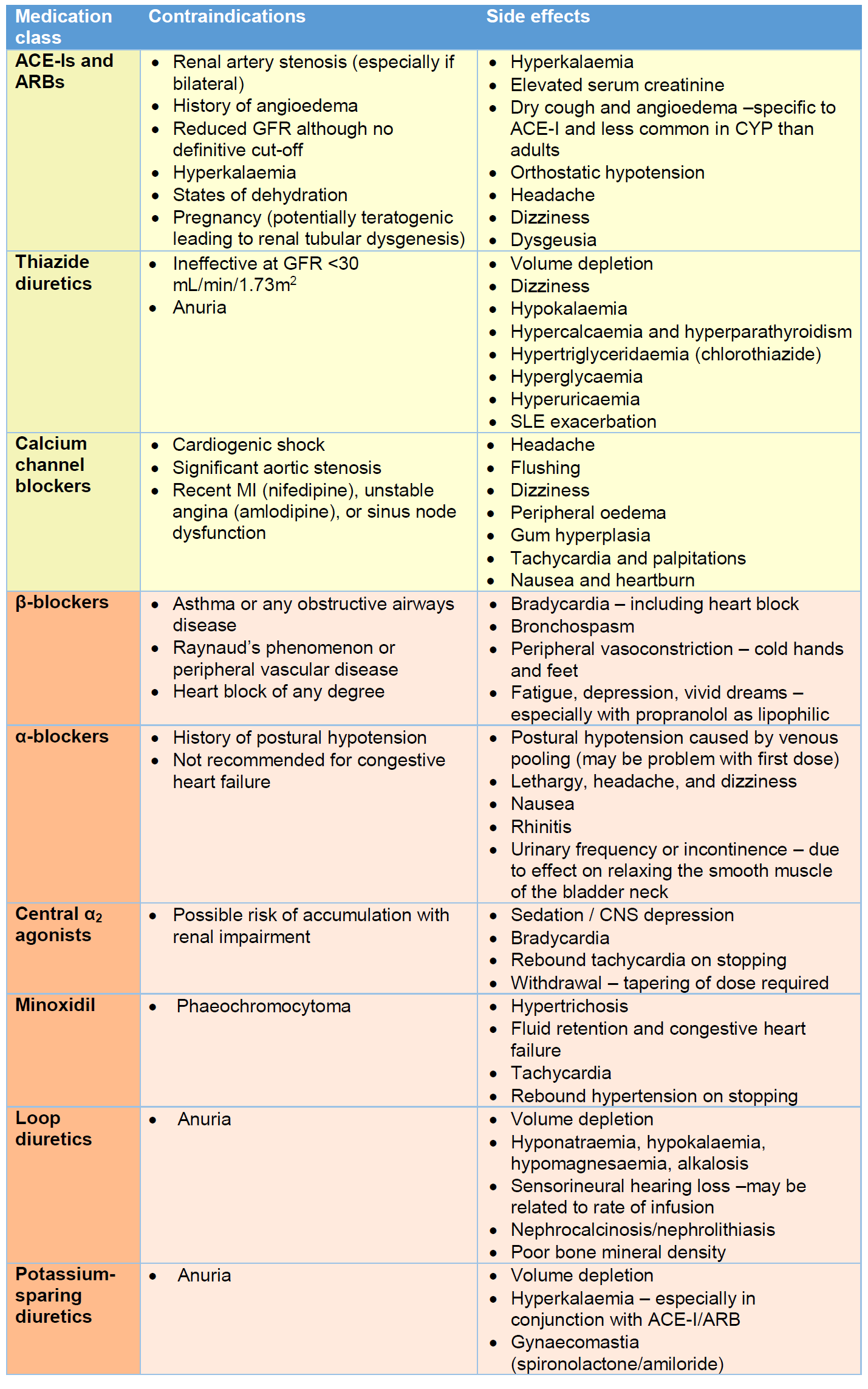
- Side effects and contraindications for the medications listed in this section are summarised in Table 4.
- First-line therapy in CYP would generally be with an angiotensin converting enzyme inhibitor (ACE-I), angiotensin receptor blocker (ARB), thiazide diuretic, or calcium channel blocker (CCB) (4). First-line agents may be used in combination, although ACE-Is and ARBs are not typically used together.
- ACE-Is/ARBs have a renoprotective effect in CKD related to their ability to reduce proteinuria (7). As such, they may be considered as first-line in those with CKD provided there are no contraindications (Table 4). In this circumstance, close monitoring of renal function will be required, particularly at the time of initiation, and after any dose adjustment. ACE-Is may be short-acting (e.g., captopril, which is given 2-3 times per day) or longer-acting (e.g., enalapril, lisinopril or ramipril; which are given once daily). If an ACE-I is associated with side effects of cough and/or angioedema (postulated to be related to inhibition of bradykinin degradation), then switching to an ARB (e.g., candesartan or losartan) may be of benefit. In the UK, ARBs are licensed for use in those ≥6 years, although candesartan is FDA approved for those ≥1 year and evidence suggests efficacy in this age group (8). On starting an ACE-I/ARB, the lowest dose for weight should ideally be chosen. It may be advisable to give a ‘test dose’ in the hospital setting and monitor BP every 15 minutes for 2 hours thereafter to monitor for severe hypotension. Renal function should be checked within a week of starting the drug, and after any dose increment, to monitor for any increase in serum creatinine and potassium.
- Thiazide diuretics (e.g., chlorothiazide and bendroflumethiazide) can be used as first-line or adjunct agents in patients with oedema and fluid overload. Chlorothiazide is available as a liquid formulation whereas bendroflumethiazide is only available as a tablet (which can be dispersed in water). They work well in combination with ACE-Is/ARBs and β-blockers (9). For maximal effect, dietary salt restriction should be adhered to. They do not work in low GFR states, or in anuria, as they need to be delivered to the distal tubule to exert their effect.
- CCBs (e.g., amlodipine or nifedipine MR) are generally well-tolerated and effective in CYP (10). They may be considered as first-line agents in those with advanced renal impairment (where ACE-Is/ARBs are contraindicated), hyperkalaemia, sexually active females who may become pregnant, and in those without evidence of oedema and fluid overload. In adults, those of black ethnicity may respond better to CCBs and thiazide diuretics than ACE-Is/ARBs (and β-blockers) (11). Due to limited data, it remains unclear whether this also applies to CYP.
- Adjunct agents should be considered if there is failure to respond to first-line therapies. These include β-blockers, α-blockers, central α2 agonists (e.g., clonidine), direct vasodilators (e.g., minoxidil), and other types of diuretics (i.e., loop and potassium-sparing). The choice of adjunct agents should be guided by the patient’s history and clinical status and should be discussed with the nephrology team.
- β-blockers may be short- (e.g., propranolol) or longer-acting (e.g., atenolol, bisoprolol, and metoprolol). Propranolol is non-selective for both β₁- and β₂-adrenoceptors. Atenolol is selective for the β₁-adrenoceptor (i.e., cardioselective).
- α-blockers may be short- (e.g., prazosin) or longer-acting (e.g., doxazosin). Those that affect BP are selective to α₁-adrenoceptors. Certain α-blockers (e.g., doxazosin) may also be used for the management or lower urinary tract symptoms due to a synergistic effect on relaxing the smooth muscle of the bladder neck.
- Clonidine is generally reserved for those unresponsive to ≥2 ‘standard’ antihypertensive agents (4). It acts as an α2-adrenergic receptor agonist in the CNS, resulting in decreased sympathetic outflow and increased firing of the vagal nerve. This leads to reduced heart rate, reduced total peripheral vascular resistance, reduced venous return, and reduced BP. Clonidine is available in oral, IV and transdermal preparations.
- Minoxidil is an adenosine 5'-triphosphate-sensitive K+ channel opener that leads to direct vasodilatation. It is generally considered a ‘last line’ antihypertensive. There is limited randomised controlled trial data on its use in CYP but evidence suggests it is effective in conjunction with a β-blocker and a diuretic (to help alleviate potential side effects of tachycardia and fluid/salt retention respectively) (12).
- Loop (e.g., furosemide, bumetanide) and potassium-sparing (e.g., spironolactone and amiloride) diuretics may be considered as adjunct agents but attention should be paid to potential electrolyte disturbances. Loop diuretics may still show efficacy in low GFR states (unlike thiazide diuretics) but higher doses may be needed. This must be balanced with the risk of inducing ototoxicity.
Table 4 – Contraindications and side effects for common classes of antihypertensive medications (13). This list is not exhaustive and the BNFC should always be consulted for dosing information, and more detailed side effect profiles. First-line therapies are in the yellow boxes, and adjunct agents in orange.
- Management of hypertensive emergency (hypertension with features of target-organ damage e.g., hypertensive encephalopathy, congestive heart failure, acute kidney injury, papilloedema and/or retinal haemorrhage):
- Support airway, breathing and circulation.
- If acute seizure occurs, then mange as per the APLS algorithm (‘Management of the convulsing child’) with first-line treatment being benzodiazepines.
- Provided that there are no neurological/neurosurgical contraindications, the next step is to lower systolic BP gradually (1/3rd reduction in the first 12 hours, then further 1/3rd over the next 12 hours, then last 1/3rd over the next 24 hours) towards the target systolic BP goal (e.g., 90th-95th centile). This should be done in the HDU/PICU setting, ideally with invasive BP assessment via an arterial line. If BP drops precipitously then IV sodium chloride 0.9% can be administered to counteract this.
- If BP is known to have become acutely elevated in a haemodialysis patient (following excessive fluid and salt intake) then BP can probably be brought down more rapidly than the proposed timeline suggested above.
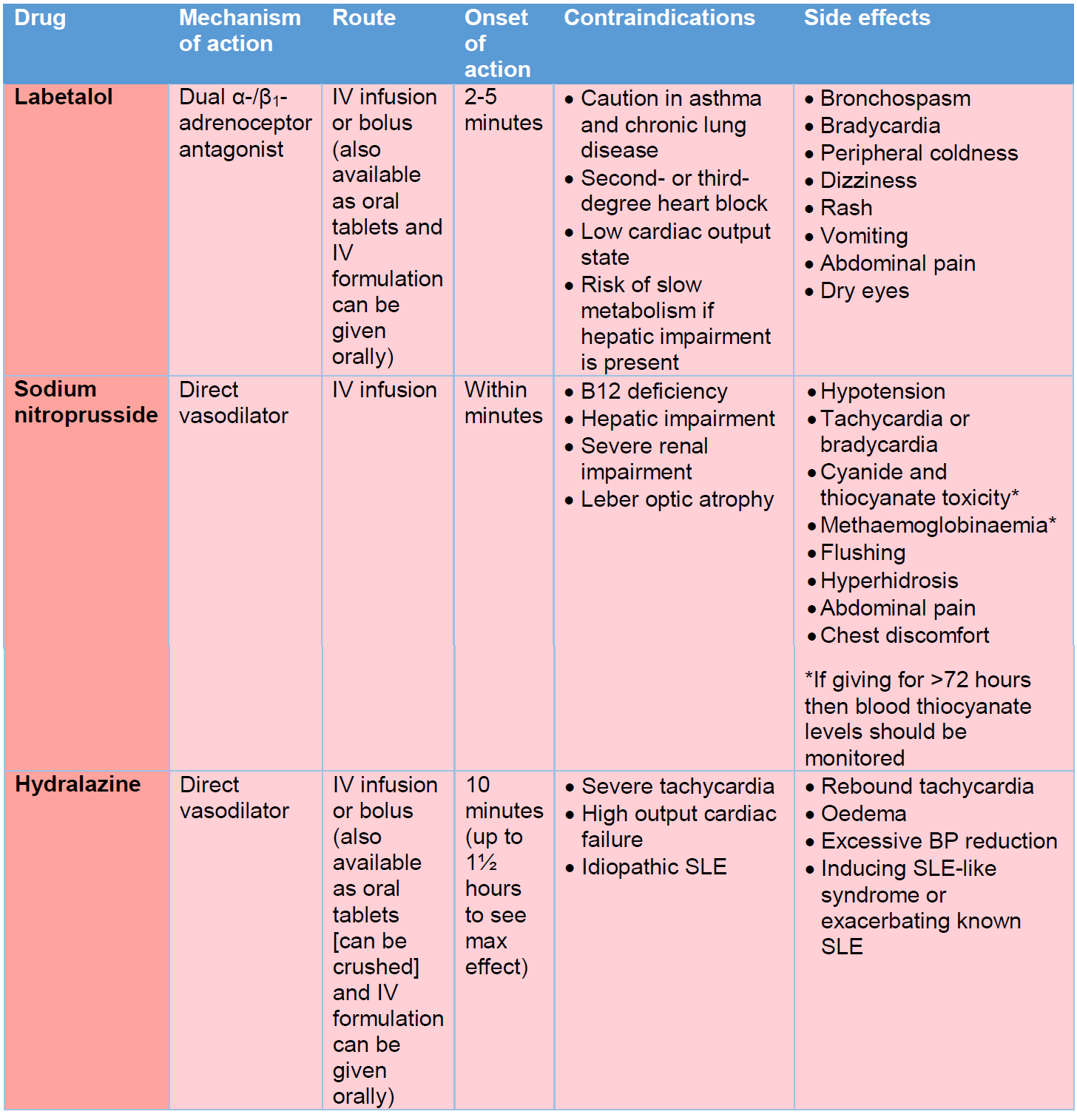
- IV labetalol would usually be considered first-line in this instance as it is easy to titrate and has a relatively short half-life of 4-8 hours.
- If there is evidence of volume overload, then a loop or thiazide diuretic should be considered. If there is significant renal impairment with oligoanuria then continuous renal replacement therapy (CRRT) may be needed to aid with fluid removal.
- If BP does not respond following the first hours of commencing the IV labetalol infusion (with incremental dosing) then a second agent should be considered. This would typically be an infusion of sodium nitroprusside (preferential) or hydralazine.
- Medications for the management of hypertensive emergency are listed in Table 5. Dose ranging should be sought from the BNFC. An additional option, not covered in Table 5, would be IV nicardipine.
- An acute cause for hypertensive emergency should be sought with baseline investigations being carried out urgently.
Table 5 – Medications for the management of hypertensive emergency.
- Management of acute severe hypertension / hypertensive urgency without evidence of emergency (i.e., stage 2 hypertension without evidence of target-organ damage):
- If hypertension is assumed to be secondary to an acute process (e.g., acute glomerulonephritis, or secondary to medications such as steroids) then consider whether diuretic therapy may be of use. If there is no evidence of fluid overload, then oral nifedipine or bolus doses of IV hydralazine may be considered to acutely lower BP although they both risk inducing a profound drop in BP.
- Immediate-release oral nifedipine is short-acting with a half-life of 1.4-4 hours. It has a rapid effect on lowering BP when given orally (unless given in the modified release preparation). The short-acting formulations come as either a solution (20 mg/mL) or as capsules (5 mg and 10 mg). The capsules should be bitten and swallowed to release the liquid if rapid response is desired. Alternatively, liquid from the capsules can be drawn up into a syringe to allow titration of the dose (if 5 mg or 10 mg is not the desired dose). There is a risk of inducing a profound drop in BP so starting with a low dose is recommended. We would recommend starting with a dose lower than that recommended in the BNFC, such as 100 micrograms/kg (max dose 10 mg), and monitoring response closely. Rebound hypertension may occur as the drug’s effect wears off. Dose can be repeated 4 hourly.
- IV hydralazine can be given as a bolus to acutely lower BP. Half-life is similar to that of nifedipine. There is a significant inter-individual response to therapy. Side effects are as listed in Table 5. Again, we would recommend starting at a low dose (i.e., 100-150 micrograms/kg for those 1 month-11 years, or 5 mg for those 12-17 years) and titrating according to response. Dose can be repeat 4 hourly.
- Early consideration should be given to starting a longer-acting antihypertensive if hypertension is likely to persist, rather than trying to control BP with intermittent dosing of short-acting agents.
- Special circumstances:
- Phaeochromocytoma:
- Initial treatment should be with an α-blocker. Traditionally, phenoxybenzamine (a non-selective, irreversible α-blocker) was the drug of choice but evidence suggests that selective α₁-blockers (e.g., doxazosin) are equally effective/safe. Once adequate α-blockade has been achieved, a β-blocker (typically propranolol or metoprolol) can be added to achieve heart rate control. Starting a β-blocker before an α-blocker is contraindicated as unopposed α-mediated vasoconstriction can lead to hypertensive crisis and pulmonary oedema.
- Monogenic forms of hypertension:
- Liddle syndrome – use of potassium-sparing diuretics such as amiloride or triamterene.
- AME – use of aldosterone antagonists such spironolactone or eplerenone, along with amiloride.
- GRA – use of potassium-sparing diuretics such as amiloride or triamterene.
- Gordon syndrome – use of a thiazide diuretic.
- Phaeochromocytoma:
- If primary hypertension is suspected, plot height, weight, and BMI. In such instances, lifestyle modification should be implemented before referral to nephrology.
- Check that the measurements are correct:
- Hypertension confirmed on at least 3 separate readings (all taken from upper limb).
- Patient’s BP centiles should be documented clearly based on gender/age/height (see Appendices 1 and 2).
- Ensure patient settled at time of assessment.
- Oscillometric readings should be confirmed manually if elevated
- In young children, this may require admission to day unit, such as Ward 1C, for serial measurements to be taken over several hours.
- If unable to assess BP reliably then discuss with nursing staff on 3C Renal to advise on Doppler assessment of BP (and provision of Doppler monitor).
- If possible, reduce or stop any drugs that may be contributing to elevated BP.
- Thorough examination of the CYP including for any evidence of neurocutaneous syndrome and full cardiac examination.
- Where reasonable, primary investigations should have been conducted, including Doppler renal ultrasound if possible, before referral to nephrology.
- Refer urgently if evidence of target-organ damage or renal involvement (acute kidney injury, proteinuria/haematuria).
- If there are any cardiac concerns, then a chest x-ray and echocardiogram should be performed urgently before referral to nephrology.
- If there are any acute neurological concerns in the context or hypertension, then the PICU team should be jointly consulted alongside nephrology.
- Lurbe E, et al. 2016 European Society of Hypertension guidelines for the management of high blood pressure in children and adolescents. J Hypertens. 2016;34(10):1887-920.
- Soergel M, et al. Oscillometric twenty-four-hour ambulatory blood pressure values in healthy children and adolescents: a multicenter trial including 1141 subjects. J Pediatr. 1997;130(2):178-84.
- Wühl E, et al. Distribution of 24-h ambulatory blood pressure in children: normalized reference values and role of body dimensions. J Hypertens. 2002;20(10):1995-2007.
- Flynn JT, et al. Clinical practice guideline for screening and management of high blood pressure in children and adolescents. Pediatrics. 2017;140(3).
- Matsuoka S, et al. White coat effect and white coat hypertension in pediatric patients. Pediatr Nephrol. 2002;17(11):950-3.
- Sacks FM, et al. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. DASH-Sodium Collaborative Research Group. N Engl J Med. 2001;344(1):3-10.
- Wühl E, et al. Antihypertensive and antiproteinuric efficacy of ramipril in children with chronic renal failure. Kidney Int. 2004;66(2):768-76.
- Schaefer F, et al. Efficacy, safety and pharmacokinetics of candesartan cilexetil in hypertensive children from 1 to less than 6 years of age. J Hypertens. 2010;28(5):1083-90.
- Berenson GS, et al. Combined low-dose medication and primary intervention over a 30-month period for sustained high blood pressure in childhood. Am J Med Sci. 1990;299(2):79-86.
- Flynn JT, et al. Treatment of hypertensive children with amlodipine. Am J Hypertens. 2000;13(10):1061-6.
- James PA, et al. 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8). JAMA. 20 14;311(5):507-20.
- Strife CF, et al. Minoxidil for control of acute blood pressure elevation in chronically hypertensive children. Pediatrics. 1986;78(5):861-5.
- The BNF for children. Drug Ther Bull. 2005;43(11):84-5.
Last reviewed: 23 January 2023
Next review: 31 January 2028
Author(s): Douglas Stewart, Consultant Paediatric Nephrologist; Angela Lamb, Paediatric Renal Pharmacist; Peter Schulga, ST8 Paediatrics (Nephrology GRID)
Version: 3
Approved By: Paediatric & Neonatal Clinical Risk & Effectiveness Committee
Document Id: 498