Investigation & Management of chemotherapy & radiotherapy induced diarrhoea & constipation in Haematology/Oncology (non-HSCT) patients
Objectives
Diarrhoea
Diarrhoea is defined as an increased frequency of bowel movements that are loose in consistency. It can be a side effect of Systemic Anti-Cancer Therapy (SACT) and can be debilitating and even life threatening, due to fluid loss and electrolyte imbalance and therefore the impact of severe diarrhoea should not be underestimated.
SACT drugs that are cited as commonly producing diarrhoea are 5-fluorouracil (5-FU), Methotrexate, Irinotecan and Taxanes (Docetaxel, Paclitaxel). However, a wide range of cytotoxic drugs, including monoclonal antibodies and hormonal treatments, and other medicines used in supportive care can also cause diarrhoea. This includes antibiotics and ciclosporin (although it should be noted that almost all drugs will include diarrhoea as a potential side effect). Cancer treatment may also cause diarrhoea indirectly:
- Infections associated with neutropenia
- Graft versus host disease of the gut following stem cell transplantation
- Radiotherapy
SACT-induced diarrhoea may have a dramatic impact on a patient’s quality of life, physical and emotional wellbeing, and invariably increases patient costs. There may be associated abdominal pain, cramping, proctitis, and anal or peri-anal skin breakdown, which in turn can lead to weight loss, malnutrition, sleep disturbance & depression.
This purpose of this guideline is to support medical and nursing staff in managing SACT-induced diarrhoea through advice on:
- assessment tools for grading diarrhoea
- the management of different grades of diarrhoea
- minimising morbidity and maximising a patient’s quality of life whilst on treatment
- reducing the need for treatment modification and delay of chemotherapy treatment
- ensuring adequate reporting of high grade toxicity to Clinical Trial Coordinators for discussion at multi-disciplinary meetings
NB: New agents, monoclonal antibodies or therapies used in Phase I & II clinical trials may have potential side effects and specific monitoring requirements that are not covered in this guideline. Staff should contact the Principle Investigator, Research Nurse or Schiehallion Pharmacists in these cases.
Constipation
Chemotherapy-induced constipation (CIC) is recognised as being a mixture of reduced frequency of bowel action and increased stool consistency; however, the mechanisms of CIC are very poorly defined.
Chemotherapy drugs known to cause constipation are the vinca alkaloids, platinums, thalidomide and hormonal agents, but there is very few published literature on true incidence or mechanism. Potential ways in which chemotherapy can alter the balance of normal gut function and lead to constipation are:
- Decreased oral intake (dehydration)
- Decreased motility (increases time for reabsorption to occur)
- Automatic neuropathy
- Increased reabsorption
- Blockage
- Overtreated diarrhoea
- Anti-nauseants
- Analgesics
- Decreased exercise
Constipation in patients with cancer is often secondary to drugs that are given to control other chemotherapy, or due to cancer-induced symptoms (such as anti-emetics for nausea and vomiting and opiods for pain) rather than to the chemotherapeutic agents themselves.
Chemotherapy-induced constipation is managed as per the GG&C guidelines (see section 2.2).
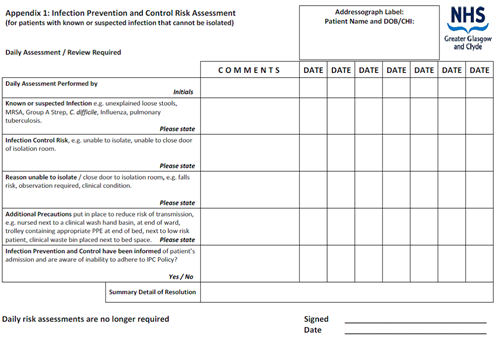
2.1 Infection Control Care Plan for Loose Stools (See Appendix 1)
Any member of qualified medical staff can investigate and manage patients with diarrhoea.
4.1 Stool sample pot or universal container
4.2 Patient / carer advice (see Appendix 2)
4.3 Grading Assessment & Adverse Event Tool (see Appendix 3)
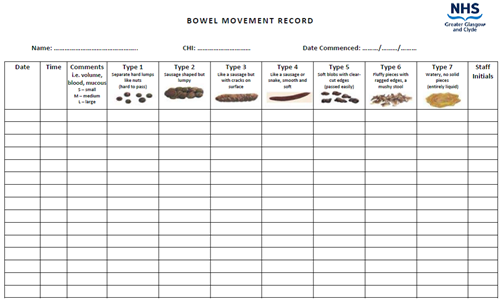
4.4 Bowel Movement Record (see Appendix 4)
5.1 Patient / Parent / Carer Information & Education:
Patient / carer information is central to the management of SACT-induced diarrhoea, including the possible causes (infection or chemotherapy side effect) and the potential for life threatening dehydration, particularly in babies and young children.
Before starting chemotherapy patients and/or parents should be informed that diarrhoea may occur and what action to take should it do so (see Appendix 2). This should be recorded in the patient casenotes.
Patients / carers will require fluid and nutrition advice in order to maintain satisfactory hydration and nutritional status. A low residue diet with high fluid intake may be appropriate.
If the diarrhoea occurs only after lactose containing foods a secondary lactose intolerance may be the cause and a trial of a lactose free diet may be appropriate. Where this is suspected it should be discussed with the dietitian.
Patients / carers must be informed that children with poor fluid intake and diarrhoea must be presented to the Schiehallion Daycare Ward or their local shared care hospital for assessment.
5.2 Pre-Chemotherapy Treatment Assessments:
Accurate pre-chemotherapy assessment is essential to enable variation from the patient’s baseline to be detected. The following should be recorded for all patients:
- Weight in kilograms
- FBC and biochemistry
- Usual bowel habit
- Patient’s use of bowel medications, e.g. laxatives
5.3 Investigations:
After obtaining a detailed history from the patient, the doctor should inspect the bowel movements if possible, or speak to a nurse who has done so.
The following should be noted in the casenotes:
- Frequency of stooling
- Volume of each bowel movement and total daily volume
- Colour of faecal material
- Consistency of faecal material – liquid stool is defined as a stool which conforms to shape of container
- Odour
- Presence of blood
- Presence of tissue fragments
- Associated symptoms e.g. nausea, vomiting, abdominal pain
To confirm whether the diarrhoea is related to SACT or infection, fresh stool specimens should be sent to the Microbiology Laboratory testing and should include:
- Routine culture for standard enteric pathogens
- Culture for ESBL’S and VRE
- Clostridium difficile will be tested in symptomatic patients aged 2-17 years on liquid stools if negative for enteric pathogens and viral PCR negative.
Stool specimens should also be sent to Virology, WoSSVC, GRI
- PCR test for Adenovirus and Rotavirus and Gastroenteritis PCR
In acute onset diarrhoea with liquid stools, Norovirus or Astrovirus should be suspected and screened weekly by Faeces Viral PCR screen (includes Norovirus, Astrovirus Rotavirus, Adenovirus and Sapovirus) and Cryptosporidium PCR (send to Parasitology Reference Laboratory, SPDRL, GRI). Other pathogens such as Giardia should be considered and appropriate samples sent for investigation.
All patients with microbiological pathogens in their stool should be isolated until they have been shown to be negative on at least 2 consecutive specimens. For viral pathogens repeat testing is not helpful and patients should be isolated until symptoms have resolved.
In patients in whom no infective agent has been isolated from initial culture and an infective cause of diarrhoea remains likely, 3 stool samples taken on consecutive days should be submitted. Further sampling after that point is unlikely to yield additional results and should not be performed. If necessary, please discuss this with the Consultant.
Patients should be investigated by a rectal biopsy (plus or minus an upper GI endoscopic biopsy) which can be organised through the gastroenterology service. Tissue specimens for virology should be sent in virus transport medium for EBV, CMV, Adenovirus PCR, Gastroenteritis PCR and Enterovirus PCR testing to WoSSVC, GRI.
5.4 Toxicity Management:
Medical and nursing management of all patients with SACT-induced diarrhoea should:
- Ensure toxicity assessment prior to each cycle of chemotherapy
- Eliminate other potential causes of diarrhoea where possible without delaying treatment, such as:
- infection
- use of laxatives
- constipation overflow
- concurrent drugs, such as antibiotics
- progressive disease
- Explain likely cause of diarrhoea to patient / carer. Explain treatment plan. Provide reassurance and support. Educate regarding personal care.
- Ensure optimum hygiene care to anal and peri-anal areas (and/or stoma site). Collaborate with tissue viability service if the patient’s skin becomes excoriated particularly for babies still in nappies.
- Ensure caregivers wear gloves when providing personal care to prevent the risk of cross-infection.
- Ensure anti-diarrhoea agents are given as prescribed, or that carers who are self-medicating understand the medicines and treatment plan.
- Monitor and record diarrhoea & associated symptoms (document on Bowel Movement Record chart – see Appendix 4:
- Frequency
- Volume
- Colour
- Consistency
- Presence of fresh blood / melaena
- Change in smell
- Abdominal cramping / pain
- Rectal bleeding
- Nausea/vomiting
- Monitor and record effects of anti-diarrhoea agents and other interventions, e.g. skin care, analgesia.
- Observe and report signs of dehydration:
- Low urine output
- Dry mucous membranes
- Sunken eyes / fontanel, absence of tears
- Poor tissue turgor
- Negative fluid balance
- Decreased peripheral perfusion
- Deep breathing
- High urea
- Low pH
- Large base deficit
- Observe and report signs of low sodium levels:
- Tiredness
- Disorientation
- Headache
- Muscle Cramps
- Nausea - severely low sodium can lead to seizures or coma. Severely low potassium can cause cardiac arrhythmias.
5.5 Grading Specific Management:
Where relevant see the patient’s specific clinical trial protocol or national treatment guideline for grading criteria or use Appendix 3 where there is no relevant protocol/guideline.
5.6 SACT Drug Specific Management - Irinotecan:
- Early diarrhoea starts during or within 24hrs of receiving Irinotecan and is cholinergic in nature. It is associated with symptoms of sweating, stomach cramps, watering eyes, blurred vision, dizziness, feeling unwell, and excessive mouth watering.
- Experience to date suggests that early diarrhoea is not a major problem. Early diarrhea may be accompanied by abdominal cramps and other cholinergic symptoms. Should treatment be necessary atropine is recommended at a dose of 0.02mg/kg (max of 0.25 mg IV or subcutaneous). If this happens, then the patient should receive prophylactic atropine at the same dose as noted above for subsequent courses of Irinotecan.
- Late onset diarrhoea – starts more than 24hrs after starting an Irinotecan infusion. Loperamide should be given according to the following schedule until a normal pattern of bowel movement returns. Oral rehydration should be given in addition throughout the episode of diarrhea as follows:
- >= 43kg: 4mg after first loose stool. Subsequently 2mg every 2 hours (2mg. every 4H at night)
- 30 - 43kg: 2mg after first loose stool. Subsequently 1mg every 2 hours (2mg. every 4H at night)
- 20 - 30kg: 2mg after first loose stool. Subsequently 1mg every 3 hours (2mg. every 4H at night)
- 13 - 20kg: 1mg after first loose stool. Subsequently 1mg. every 3 hours (1mg. every 4H at night
- < 13kg: 0.5mg after first loose stool. Subsequently 0.5mg every 3 hours (0.5mg. every 4H at night)
- If a patient needs to take Loperamide they and/or their carers should be counselled to maintain close contact with shared local hospital/shared care treatment centre or the Schiehallion Daycare Ward (2B) / Ward 2A, and certainly to report if the diarrhoea has not resolved within 48 hours.
- Loperamide should not be given prophylactically, even in patients who experienced delayed diarrhoea in previous cycles.
- Where the delayed diarrhoea is unresponsive to Loperamide, a trial of Cefixime may be appropriate. Cefixime reduces bowel colonisation by organisms that may reactivate the active metabolite of Irinotecan excreted in the bile, leading to local toxicity. The dose is 8mg/kg/day (Max: 400mg) for two days before Irinotecan and through the course and seven days after Irinotecan is completed.
Infection Control Care Plan for Loose Stools

Infection Prevention and Control Risk Assessment
Patient / Parent / Carer Advice
Whilst your child is at home on chemotherapy or post-radiotherapy it is important that you monitor their health. Most importantly are fever (make contact urgently if temperature is above 38o) and bowel movements.
Please call the Schiehallion Daycare Ward (2B) on 0141 452 4476 / 4476 during the hours of 08:30 to 19:00 (Monday to Friday) or the Schiehallion Ward (2A) on 0141 452 4450 / 4451 outside of these hours/days for advice if your child develops diarrhoea at home.
If they present with any of the following, please call immediately:
- Fever associated with diarrhoea
- Abdominal cramps / pain / bloating (especially if receiving vinca-alkaloids ie Vinblastine, Vinorelbine, Vincristine or Vindesine) as the diarrhoea may relate to constipation overflow)
- Dizziness
- Blood in faeces
- Inability to drink adequate amounts of fluid
- Low urine output, dry mouth, sunken eyes or sunken fontanel in a baby
It is important to rule out infection when a patient develops diarrhoea and therefore advice should always be sought before commencing any anti-diarrhoeal medication. If infection is ruled out the following guidance will be recommended:
- Drink plenty of fluids (clear fluids are best ie water, diluting juice. Avoid milk / milk-based drinks)
- Eat small amounts of bland low fibre foods (bananas, rice, noodles, white bread, skinned chicken/turkey, white fish etc) until diarrhoea resolves
- Avoid greasy/fried foods, raw vegetables, fruit, whole grain breads/cereals, lactose containing products, caffeine, spicy foods and gas-forming foods (beans, cabbage, broccoli) and fizzy drinks until the diarrhoea resolves
- Stop any laxatives
- Monitor temperature and report pyrexia
- Monitor diarrhoea and report immediately if there is any increase in stools, signs of dehydration, low urine output, dry mouth or sunken eyes / sunken fontanel in a baby
Grading Assessment Tool & Adverse Event Tool
Diarrhoea Grading Assessment:
|
GRADE |
CRITERIA |
MANAGEMENT |
|
1 |
2 – 3 stools per day |
Commence loperamide (Imodium):
|
|
2 |
4 – 6 stools per day or moderate cramps |
As Grade 1:
|
|
3 |
7 – 9 stools per day or severe cramps |
Withhold chemotherapy:
|
|
4 |
>10 stools per day, bloody, parenteral support required |
Urgent medical review:
|
Common Terminology Criteria for Adverse Events:
|
TOXICITY |
GRADE 1 |
GRADE 2 |
GRADE 3 |
GRADE 4 |
GRADE 5 |
|
Diarrhoea (without stoma) |
Increase of <4 stools per day |
Increase of <4 – 6 stools |
Increase of >7 stools per |
Requires intensive |
Death |
|
Diarrhoea (with stoma) |
Mild increase in loose watery output |
Moderate increase in loose watery output (>3 – 4) |
Severe increase in output, interfering with normal |
Requires intensive |
Death |
Last reviewed: 01 May 2021
Next review: 31 May 2023
Author(s): Dr Milind Ronghe
Version: 3
Approved By: Schiehallion Unit Clinical Governance Group

