Nutrition, enteral: introducing and establishing (PICU) & management of "High Risk Abdomen" patients
exp date isn't null, but text field is
Objectives
- To provide clear guidance to nursing and medical staff on introducing and establishing enteral nutrition (gastric and jejunal) in paediatric critical care patients.
- To standardise time taken to establish enteral nutrition.
- To provide clear guidance on the measuring of GRVs and how these should be interpreted.
- To provide clear guidance to PICU staff on the nutritional care management of ‘High Risk Abdomen’ patients and thereby ensure consistency of care provision.
This guideline should be interpreted in conjunction with the NHSGG&C PICU Guideline on Administration of Medication via Enteral Feeding Tubes 2009 and NHSGG&C PICU Guideline on Blind Bedside Jejunal Tube Insertion 2009.
Scope
This guideline applies to all patients in Ward 1D, Paediatric Intensive Care Unit (PICU) within the Royal Hospital for Children in Glasgow who require enteral nutrition.
Audience
All healthcare professionals in PICU involved in the provision of nutrition should be familiar with this guideline.
Optimal provision of nutrition support and accurate assessment of energy expenditure should form an integral component of paediatric critical care (Mehta et al, 2009a). Nutritional status has a profound effect on the metabolic response to injury and can impact significantly on patient outcome (Mitchell et al, 1994) including length of ventilated days and overall length of PICU stay (Larsen et al, 2008). The goal of nutrition support in the critical care setting aims to augment the short term benefits of the paediatric stress response while minimising the long term harmful consequences associated with undernutrition.
It is widely accepted that enteral nutrition is the preferred method of feeding due to its physiological advantages of maintaining gut integrity and reducing infectious complications compared with parenteral nutrition. There are numerous studies which suggest that early implementation of nutritional support (within 24hrs) is associated with improved clinical outcomes, shorter length of stay, decreased infection rates and enhanced immune function (Petrillo-Albarnano et al 2006). This supports the theory that systematic initiation of enteral nutrition may improve patient outcomes in critical illness.
Providing nutritional care in critically ill children poses a number of challenges including fluid restriction, digestive intolerance and interruptions in enteral feed delivery for diagnostic and therapeutic procedures (Lambe et al, 2007, Mara et al, 2004) resulting in a failure to deliver estimated energy requirements (Gentles et al. 2014, Mara et al. 2014, Cohen et al 2000 and de Neef et al, 2007). Failure to provide nutritional intake equal to or exceeding the predicted basal metabolic rate (PBMR) is associated with higher mortality and morbidity rates (Briassoulis et al, 2001).
The lack of controlled research and clinical trials in critically ill children has resulted in an absence of widely accepted evidence based guidelines for optimal nutritional management in this group (Van der Kuip et al, 2004). However many UK PICU’s have implemented local guidelines for introducing and establishing enteral nutrition in their critically ill populations. It has been reported that this has resulted in more rapid initiation of enteral feeding, shortened the time taken to reach nutritional goals and improved the tolerance of enteral feeding (Gentles et al. 2014, Petrillo—Albarnano et al, 2006 and Briassoulis et al, 2001).
Measurement of gastric residual volumes (GRVs) is used routinely to assess enteral feed tolerance in both adult and paediatric critical care units. Elevated GRVs are associated with sedation and catecholamine use (Mentec et al, 2001) and in paediatric patients are often attributed to gastroparesis. There is wide debate over the use of measuring GRVs to assess feed tolerance. Although McClave et al (2005) state that GRVs are inherently flawed as they have yet to be validated, current opinion also suggests that a GRV > 5ml/kg can be considered to be an indicator of poor feed tolerance and delayed gastric emptying (Cobb et al, 2004 and Horn et al, 2004).
A local guideline addressing both the introduction and advancement of enteral feed as well as the measurement of GRVs should improve nutritional delivery in PICU.
Initiating nutritional support in infants following cardiac surgery is influenced by a number of factors but principally the clinical assessment of cardiac output is crucial in deciding whether to initiate enteral or parenteral nutrition. Reduced cardiac output particularly when requiring inotropic support results in increased concern regarding splanchnic hypoperfusion and gut ischaemia. Patients with single ventricle physiology (i.e. HLHS following Norwood stage 1 palliation), reliant on a balanced circulation may be at increased risk of compromised splanchnic blood flow (Owens and Musa, 2009). Novel monitoring modalities have recently demonstrated impaired mesenteric/splanchnic tissue oxygenation in neonates who develop necrotising enterocolitis (NEC) (Fortune et al, 2001) and similarly in a case study of a patient with congenital heart disease who developed NEC (Stapleton et al 2007). The risk of NEC in neonates with congenital heart disease is substantial (McElhinney et al, 2000).
Following cardiopulmonary bypass Malagon et al (2005) identified that gut permeability is increased and damage to the intestinal mucosa is sustained, this once again leads to increased concern regarding the introduction and advancement of enteral nutrition in such a fragile patient group due increased concern of necrotising enterocolitis (NEC). Similarly, increased gut mucosal permeability leading to compromised intestinal integrity has been found in patients undergoing veno-arterial extracorporeal membrane oxygenation (ECMO), leading to bacterial translocation and sepsis (Kurundkar et al 2010; Piena et al, 1998). Introducing enteral nutrition in patients undergoing veno-venous ECMO has been reported to be safe in the adult population (Scott et al, 2004) and the use of enteral feeding in neonates on both VV and VA ECMO is advocated, however, the rate at which enteral feed should be advanced has yet to be examined (Piena et al, 1998, Pettignano et al, 1998 and Hanekamp et al, 2005).
A study in stable preterm neonates found that splanchnic oxygenation increases following bolus gastric feeding (Dave et al, 2009), however, in patients at risk of mesenteric ischaemia this increased oxygenation demand may further increase the risk of NEC and warrants further research. It could be argued that continuous feeding may be more appropriate than bolus, thereby reducing the demand on mesenteric oxygen consumption.
The introduction of feeding algorithms in patients with HLHS have been shown to enhance nutritional care and patient outcome (Castillo et al 2010; Braudis et al, 2009) and the introduction of such guidelines for patients identified as ‘High Risk Abdomen’ may also achieve similar improvements in patient morbidity and mortality.
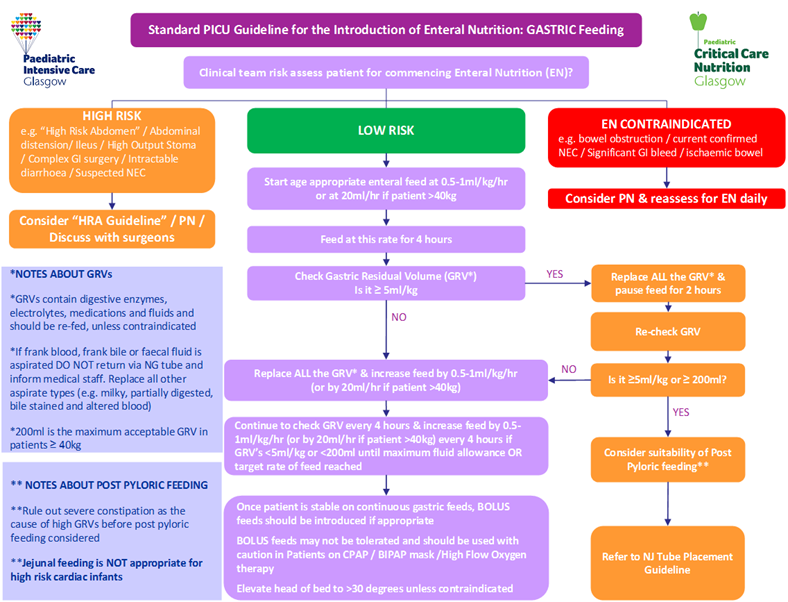
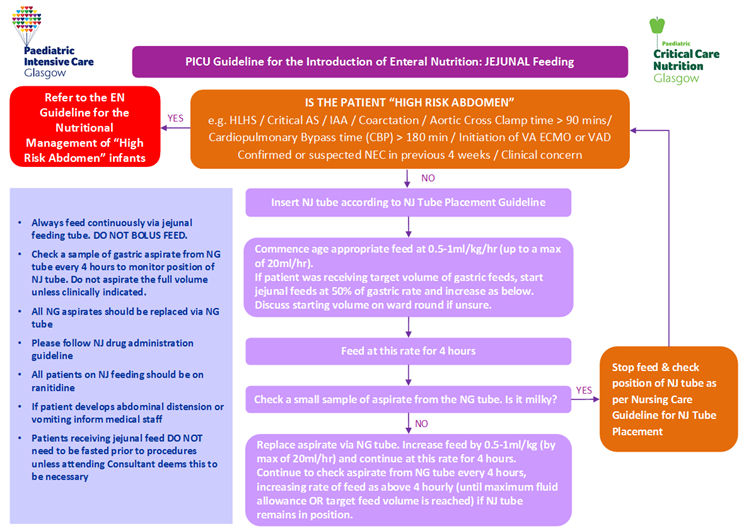
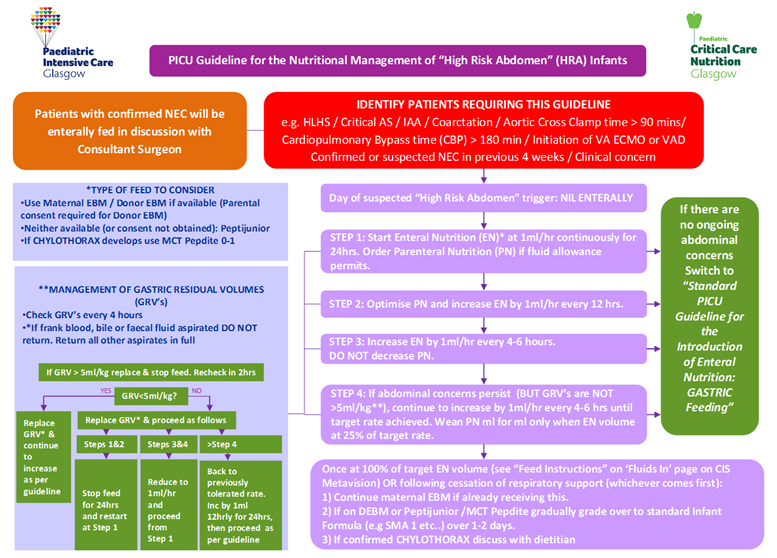
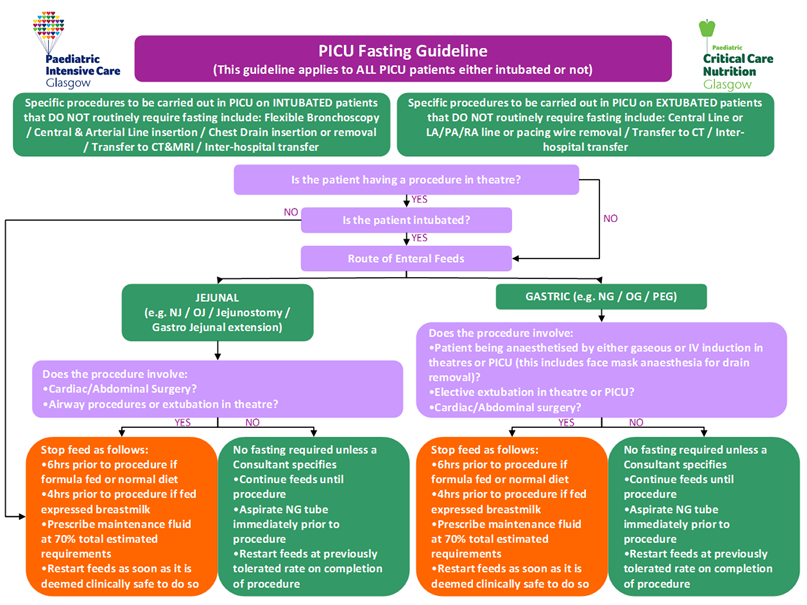
The following tables are for use in conjunction with PICU Guidelines for Introducing & Establishing Enteral Nutrition (Gastric and Jejunal). Table 1 details the initiation and advancement rates of enteral feeds. Tables 2A-C details the recommended target daily volume of enteral feed to meet nutritional requirements within given age ranges.
By using the below initiation and advancement rates patients will meet their basal metabolic rate (BMR) within 2 days of starting enteral nutrition (if fluid balance and gastrointestinal tolerance allows the increase to goal volumes detailed in Tables 2A-C).
The below feed target volumes (Tables 2(a)-2(c)) are calculated using the Schofield Predictive Formula for calculating resting energy expenditure (using median weight from the UK-WHO growth charts ages 0-4 and the UK 1990 reference for children aged >4 years), and the Scientific Advisory Committee on Nutrition (SACN), 2011 in addition to the Department of Health guidelines for EAR, 1991.
Calculations for target daily volumes are made using median weight from the UK-WHO growth charts ages 0-4 and the UK 1990 reference for children aged >4 years. Where EAR using the SACN, 2011 recommendations and the Department of Health guidelines for EAR, 1991 differed by >20% (e.g. age group 7-12 months), a mean of the two values is used to calculate target rates for EAR.
Table 1: Initiation and Advancement Rates for Enteral Nutrition
|
Weight |
Initiation Rate |
Advancement Rate |
Goal Rate |
|
0-40kg |
Start at 0.5-1ml/kg/hr (maximum of 20ml/hr) |
Increase by 0.5-1ml/kg (or maximum of 20ml/hr)every 4 hours |
|
|
>40kg |
20ml/hr |
Increase by 0.5-1ml/kg every 4 hours |
Table 2(a): Recommended Target Daily EN Volume: Infants Male & Female, age <12 months
|
Age (months) |
Feed Conc (kcal/ml) |
Feed Type |
|
||
|
|
|||||
|
0-3 |
3-7 |
Extubated or >Day 7 |
|||
|
BMR-70%EAR |
70-100%EAR |
100%EAR |
|||
|
1-2 |
0.7 |
Std Formula/EBM |
75-100 |
100-145 |
145-180 |
|
|
1 |
*Infatrini / *Infatrini Peptisorb |
50-75 |
75-96 |
96-120 |
|
3-4 |
0.7 1 |
Std Formula/EBM |
75-100 |
100-145 |
145 |
|
|
0.7 1 |
*Infatrini / *Infatrini Peptisorb |
50-75 |
75-96 |
96 |
|
5-6 |
0.7 1 |
Std Formula/EBM |
82-100 |
100-135 |
135 |
|
|
0.7 1 |
*Infatrini / *Infatrini Peptisorb |
55-70 |
70-90 |
90 |
|
7-12 |
0.7 1 |
Std Formula/EBM |
82-100 |
100-135 |
135 |
|
|
0.7 1 |
*Infatrini / *Infatrini Peptisorb |
55-70 |
70-90 |
90 |
*Infatrini /Infatrini Peptisorb should not be used in children <3 kg and should be used with caution in those <5kg. Notify dietitian if patient admitted on Infatrini/Infatrini Peptisorb.
Table 2(b): Recommended Target Daily Volumes of EN, Males >1 year old
|
Table 2(b): Recommended Target Daily Volumes of EN, Males >1 year old |
|||||
|
Age (years) |
Feed conc. (kcal/ml) |
Feed Type |
Daily Target Feed Volume (ml/kg/day) |
||
|
Day of Admission |
|||||
|
0-3 BMR-70%EAR |
3-7 70-100%EAR |
Extubated or >Day 7 100%EAR |
|||
|
1 |
1.0 |
Nutrini |
56 |
56-80 |
80 |
|
(9.6kg) |
1.5 |
Nutrini Energy |
37 |
37-53 |
53 |
|
2 |
1.0 |
Nutrini |
57 |
57-82 |
82 |
|
(12.2kg) |
1.5 |
Nutrini Energy |
38 |
38-55 |
55 |
|
3 |
1.0 |
Nutrini |
58 |
58-82 |
82 |
|
(14.4kg) |
1.5 |
Nutrini Energy |
39 |
39-55 |
55 |
|
4 |
1.0 |
Nutrini |
54-60 |
60-85 |
85 |
|
(16.3kg) |
1.5 |
Nutrini Energy |
36-40 |
40-57 |
57 |
|
5 |
1.0 |
Nutrini |
50-56 |
56-80 |
80 |
|
(18.6kg) |
1.5 |
Nutrini Energy |
33-37 |
37-53 |
53 |
|
6 |
1.0 |
Nutrini |
47-52 |
52-74 |
74 |
|
(21.0kg) |
1.5 |
Nutrini Energy |
31-35 |
35-49 |
49 |
|
7 |
1.0 |
Nutrison Standard |
45-50 |
50-71 |
71 |
|
(23.0kg) |
1.5 |
Nutrison Energy |
30-33 |
33-47 |
47 |
|
8 |
1.0 |
Nutrison Standard |
42-47 |
47-67 |
67 |
|
(26.0kg) |
1.5 |
Nutrison Energy |
28-31 |
31-45 |
45 |
|
9 |
1.0 |
Nutrison Standard |
40-44 |
44-63 |
63 |
|
(29.0kg) |
1.5 |
Nutrison Energy |
27-29 |
29-42 |
42 |
|
10 |
1.0 |
Nutrison Standard |
39-45 |
45-64 |
64 |
|
(31.5kg) |
1.5 |
Nutrison Energy |
26-30 |
30-43 |
43 |
|
11 |
1.0 |
Nutrison Standard |
37-43 |
43-62 |
62 |
|
(34.5kg) |
1.5 |
Nutrison Energy |
25-29 |
29-41 |
41 |
|
12 |
1.0 |
Nutrison Standard |
35-41 |
41-59 |
59 |
|
(38.0kg) |
1.5 |
Nutrison Energy |
23-27 |
27-39 |
39 |
|
13 |
1.0 |
Nutrison Standard |
33-39 |
39-56 |
56 |
|
(43.0kg) |
1.5 |
Nutrison Energy |
22-26 |
26-37 |
37 |
|
14 |
1.0 |
Nutrison Standard |
31-38 |
38-54 |
54 |
|
(49.0kg) |
1.5 |
Nutrison Energy |
21-25 |
25-36 |
36 |
|
15 |
1.0 |
Nutrison Standard |
30-36 |
36-51 |
51 |
|
(55.5kg) |
1.5 |
Nutrison Energy |
20-24 |
24-34 |
34 |
|
16 |
1.0 |
Nutrison Standard |
29-34 |
34-49 |
49 |
|
(60.2kg) |
1.5 |
Nutrison Energy |
19-23 |
23-33 |
33 |
|
17 |
1.0 |
Nutrison Standard |
28-34 |
34-48 |
48 |
|
(64.0kg) |
1.5 |
Nutrison Energy |
19-23 |
23-32 |
32 |
|
18 |
1.0 |
Nutrison Standard |
28-34 |
34-48 |
48 |
|
(66.2kg) |
1.5 |
Nutrison Energy |
19-23 |
23-32 |
32 |
Table 2(c): Recommended Target Daily Volumes of EN, Females >1 year old
|
Age (years) |
Feed conc. (kcal/ml) |
Feed Type |
Daily Target Feed Volume (ml/kg/day) |
||
|
Day of Admission |
|||||
|
0-3 BMR-70%EAR |
3-7 70-100%EAR |
Extubated or >Day 7 100% EAR |
|||
|
1 |
1.0 |
Nutrini |
55 |
55-80 |
80 |
|
(9.0kg) |
1.5 |
Nutrini Energy |
37 |
37-53 |
37-53 |
|
2 |
1.0 |
Nutrini |
56 |
56-81 |
81 |
|
(11.5kg) |
1.5 |
Nutrini Energy |
37 |
37-54 |
54 |
|
3 |
1.0 |
Nutrini |
55 |
55-78 |
78 |
|
(13.9kg) |
1.5 |
Nutrini Energy |
37 |
37-52 |
52 |
|
4 |
1.0 |
Nutrini |
51-57 |
57-81 |
81 |
|
(16.0kg) |
1.5 |
Nutrini Energy |
34-38 |
38-54 |
54 |
|
5 |
1.0 |
Nutrini |
47-53 |
53-75 |
75 |
|
(18.2kg) |
1.5 |
Nutrini Energy |
31-35 |
35-50 |
50 |
|
6 |
1.0 |
Nutrini |
43-49 |
49-70 |
70 |
|
(21.0kg) |
1.5 |
Nutrini Energy |
29-33 |
33-47 |
47 |
|
7 |
1.0 |
Nutrison Standard |
41-47 |
47-67 |
67 |
|
(23.0kg) |
1.5 |
Nutrison Energy |
27-31 |
31-45 |
45 |
|
8 |
1.0 |
Nutrison Standard |
39-44 |
44-63 |
63 |
|
(26.0kg) |
1.5 |
Nutrison Energy |
26-29 |
29-42 |
42 |
|
9 |
1.0 |
Nutrison Standard |
37-41 |
41-59 |
59 |
|
(29.0kg) |
1.5 |
Nutrison Energy |
25-27 |
27-39 |
39 |
|
10 |
1.0 |
Nutrison Standard |
35-43 |
43-61 |
61 |
|
(32.0kg) |
1.5 |
Nutrison Energy |
23-29 |
29-41 |
41 |
|
11 |
1.0 |
Nutrison Standard |
33-39 |
39-56 |
56 |
|
(35.9kg) |
1.5 |
Nutrison Energy |
22-26 |
26-37 |
37 |
|
12 |
1.0 |
Nutrison Standard |
31-37 |
37-53 |
53 |
|
(40.0kg) |
1.5 |
Nutrison Energy |
21-25 |
25-35 |
35 |
|
13 |
1.0 |
Nutrison Standard |
28-34 |
34-48 |
48 |
|
(46.0kg) |
1.5 |
Nutrison Energy |
32-23 |
23-32 |
32 |
|
14 |
1.0 |
Nutrison Standard |
27-32 |
32-46 |
46 |
|
(51.0kg) |
1.5 |
Nutrison Energy |
18-21 |
21-31 |
31 |
|
15 |
1.0 |
Nutrison Standard |
26-32 |
32-45 |
45 |
|
(53.0kg) |
1.5 |
Nutrison Energy |
17-21 |
21-30 |
30 |
|
16 |
1.0 |
Nutrison Standard |
26-31 |
31-44 |
44 |
|
(55.3kg) |
1.5 |
Nutrison Energy |
17-21 |
21-29 |
29 |
|
17 |
1.0 |
Nutrison Standard |
26-30 |
30-43 |
43 |
|
(57.0kg) |
1.5 |
Nutrison Energy |
17-20 |
20-29 |
29 |
|
18 |
1.0 |
Nutrison Standard |
29-30 |
30-43 |
43 |
|
(57.2kg) |
1.5 |
Nutrison Energy |
19-20 |
20-29 |
29 |
When initiating enteral feeds in PICU use Table 3 to guide you through feed choice.
|
Age / Weight Range |
Type of Enteral Feed |
|
< 12 months (or <12 months corrected age for premature infants born <37 weeks) |
|
|
1-6 years (and weight >8kg) |
|
|
> 6 years (and weight >20kg) |
|
*For all High Energy Enteral Feeds and Specialist Enteral Feeds please consult PICU Dietitian before initiating in PICU
- Braudis NJ, Curley MAQ, Beaupre K, Thomas KC, Hardiman G, Laussen P, Gauvreau K and Thiagarajan RR (2009) Enteral feeding algorithm for infants with hypoplastic left heart syndrome poststage I palliation. Pediatric Critical Care Medicine 10 (4) 460-466.
- Briassoulis G, Zavras NJ, Hatzis MT (2001) Effectiveness and safety of a protocol for promotion of early intragastric feeding in critically ill children. Pediatric Critical Care Medicine 2(2): 113-121
- Cobb BA, Waldemar CA and Ambalavanan N (2004) Gastric residuals and their relationship to necrotizing enterocolitis in very low birth weight infants. Pediatrics 113 (1) 50-53.
- Cohen J, Aharon A and Singer P (2000) The paracetamol absorption test: a useful addition to the enteral nutrition algorithm? Clinical Nutrition 19 (4) 233-236.
- Dave V, Brion LP, Campbell DE, Scheiner M, Raab C and Nafday SM (2009) Splanchnic tissue oxygenation, but not brain tissue oxygenation, increases after feeds in stable preterm neonates tolerating full bolus orogastric feeding. Journal of Perinatology 29 213-218.
- Del Castillo SL, McCulley ME, Khemani RG, Jeffries HE, Thomas DW, Peregrine J, Wellls WJ, Starnes VA and Moromisato (2010) Reducing the incidence of necrotizing enterocolitis in neonates with hypoplastic left heart syndrome with the introduction of an enteral feed protocol. Pediatric Critical Care Medicine 11 (3) 373-377
- de Neef M, Geukers VGM, Dral A, Lindeboom R, Sauerwein HP and Bos AP (2008) Nutritional goals, prescription and delivery in a pediatric intensive care unit. Clinical Nutrition 27 65-71.
- Fortune PM, Wagstaff M and Petros AJ (2001) Cerebro-splanchnic oxygenation ratio (CSOR) using near infrared spectroscopy may be able to predict splanchnic ischaemia in neonates. Intensive Care Medicine 27 1401-1407.
-
Gentles E, Mara J, Diamantidi K, Alfheeaid H, Spenceley N, Davidson M, Gerasimidis K (2014) Delivery of Enteral Nutrition after the introduction of practice guidelines and participation of dietitians in paediatric critical care clinical teams. J Acad Nutr Diet 114(12): 1974-80
- Hanekamp MN, Spoel M, Sharman-Koendjbiharie I, Peters JWB, Albers MJIJ and Tibboel D (2005) Routine enteral nutrition in neonates on extracorporeal membrane oxygenation. Pediatric Critical Care Medicine 6 (3) 275-279.
- Horn D, Chaboyer W and Schluter PJ (2004) Gastric residual volumes in critically ill paediatric patients: A comparison of feeding regimens. Australian Critical Care 17 (3) 98-103.
- Kurundkar AR, Killingsworth CR, McIIwain RB, Timpa JG, Hartman YE, He D, Karnatak RK, Neel ML, Clancy JP, Anantharamaiah GM and Maheshwari A (2010) Extracorporeal membrane oxygenation causes loss of intestinal epithelial barrier in the newborn piglet. Pediatric Research 68 (2) 128-133.
- Lambe C, Hubert P, Jouvet P, Cosnes J and Colomb V (2007) A nutritional support team in the pediatric intensive care unit: Changes and factors impeding appropriate nutrition. Clinical Nutrition 26 355-363.
- Larsen B Goonewardene L, Joffe AR, Van Aerde J, Field CJ and Clandinin MT (2008) Energy intake significantly affects hospital outcomes in infants after open heart surgery. Journal of Parenteral and Enteral Nutrition 32 (3) 317.
- Lopez-Herce (2009) Gastrointestinal complications in critically ill patients: what differs between adults and children. Current Opinion in Clinical Nutrition and Metabolic Care 12 180-185.
- Malagon I, Onkenhout W, Klok G, van der Poel PFH, Bovill JG and Hazekamp MG (2005) Gut permeability in paediatric cardiac surgery. British Journal of Anaesthesia 94 (2) 181-185.
-
Mara J, Gentles E, Alfheeid H, Diamantidi K, Spenceley N, Davidson M, Young D, Gerasimidis K (2014) An evaluation of enteral nutrition practices and nutritional provision in children during the entire length of stay in critical care. BMC Pediatrics. 14: 186
- McClave SA, Lukan JK, Stefater JA, Lowen CC, Looney SW, Matheson PJ, Gleeson K and Spain DS (2005) Poor validity of residual volumes as a marker for risk of aspiration in critically ill patients. Critical Care Medicine 33 (2) 324-330.
- McElhinney DB, Hedrick HL, Bush DM, Pereira GR, Stafford PW, Gaynor JW, Spray TL and Wernovsky G (2000) Necrotizing enterocolitis in neonates with congenital heart disease: risk factors and outcomes. Pediatrics 105 (5) 1080-1087.
- Mehta NM, Bechard LJ, Leavitt K and Duggan C (2009a) Cumulative energy imbalance in the pediatric intensive care unit: role of targeted indirect calorimetry. Journal of Parenteral and Enteral Nutrition 33 (3) 336-344.
- Mehta N, Compher C and ASPEN Board of Directors (2009b) ASPEN Clinical Guidelines: Nutrition Support of the Critically Ill Child. Journal of Parenteral and Enteral Nutrition 33 (3) 260-276.
- Mentec H, Dupont H, Bocchetti M, Cani P, Ponche F and Bleighner G (2001) Upper digestive intolerance during enteral nutrition in critically ill patients: frequency, risk factors, and complications. Critical Care Medicine 29 (10) 1955-1961.
- Mitchell IM, Davies PSW, Day JME, Pollock JCS, Jamieson MPG (1994) Energy expenditure in children with congenital heart disease, before and after cardiac surgery. The Journal of Thoracic and Cardiovascular Surgery 107 (2) 374-380.
- Owens JL and Musa N (2009) Nutrition support after neonatal cardiac surgery. Nutrition in Clinical Practice 24 (2) 242-249.
- Petrillo-Albarano T, Pettignano R, Asfaw M, Easley K (2006). Use of a feeding protocol to improve nutritional support through early aggressive enteral nutrition in the paediatric intensive care unit. Paediatric Critical Care Medicine. 7:340-344.
- Pettignano R, Heard M, Davis R, Labuz M and Hart M (1998) Total enteral nutrition versus total parenteral nutrition during pediatric extracorporeal membrane oxygenation. Critical Care Medicine 26 (2) 358-363.
- Piena M, Albers MJIJ, Van Haard PMM, Gischler S and Tibboel D (1998) Introduction of enteral feeding in neonates on extracorporeal membrane oxygenation after evaluation of intestinal permeability changes. Journal of pediatric surgery 33 (1) 30-34.
- Scott LK, Boudreaux K, Thaljeh F, Grier LR and Conrad SA (2004) Early enteral feedings in adults requiring venonvenous extracorporeal membrane oxygenation. Journal of Parenteral and Enteral Nutrition 28 (5) 295-300.
- Stapleton GE, Eble BK, Dickerson HA, Andropoulos DB and Chang AC (2007) Mesenteric oxygen desaturation in an infant with congenital heart disease and necrotizing enterocolitis. Texas Heart Institute Journal 34 (4) 442-444.
- Van der Kuip M, Oosterveld MJS, Van Bokhorst-de van der Schueren MAE, Lafeber HA and Gemke RJBJ (2004) Nutritional support in 111 pediatric intensive care units: a European survey. Intensive Care Medicine 30 1807-1813.
Last reviewed: 01 August 2016
Next review: 08 December 2022
Author(s): Emma Gentles (Dietitian), Dr Neil Spenceley (Consultant), Dr Mark Davidson (Consultant), Susan Miller (Charge Nurse)
Reviewer Name(s): Dr Neil Spenceley
Document Id: 34

